A. Magnesium chloride
B. Calcium oxide
C. Copper nitrate
D. Aluminium chloride
E. Potassium nitrate
Hint: Chemical formulas can also be created using the criss-cross method. In this approach, the numerical value of the charge of each ion is swapped, becoming the subscript for the opposing ion. Using this technique, we will write the chemical formula of the given compounds.
Complete step by step answer:
Let’s us discuss about the given compound as,
A. Magnesium chloride
It is important to note that magnesium has an atomic number of 12 and a valency of 2. It means it has two electrons in the outermost shell for bonding. The atomic number of chlorine is 1717 and has 7 electrons in the outermost shell. This indicates that it requires one additional atom for bonding. Therefore, we will use chlorine atoms to bond with one magnesium atom. We can utilize the criss-cross method for this compound as follows:
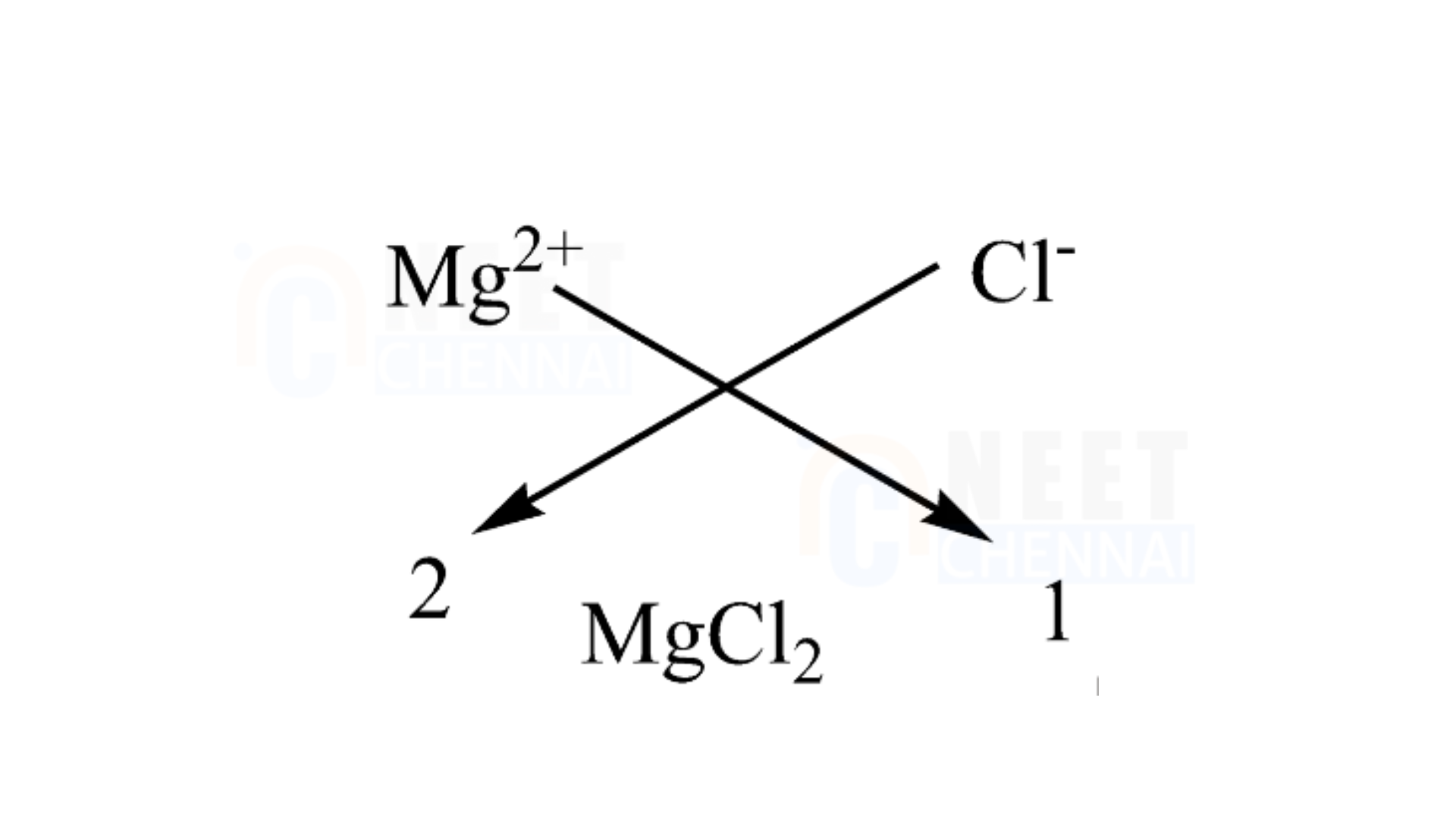
Therefore, the chemical formula of magnesium chloride is MgCl2.
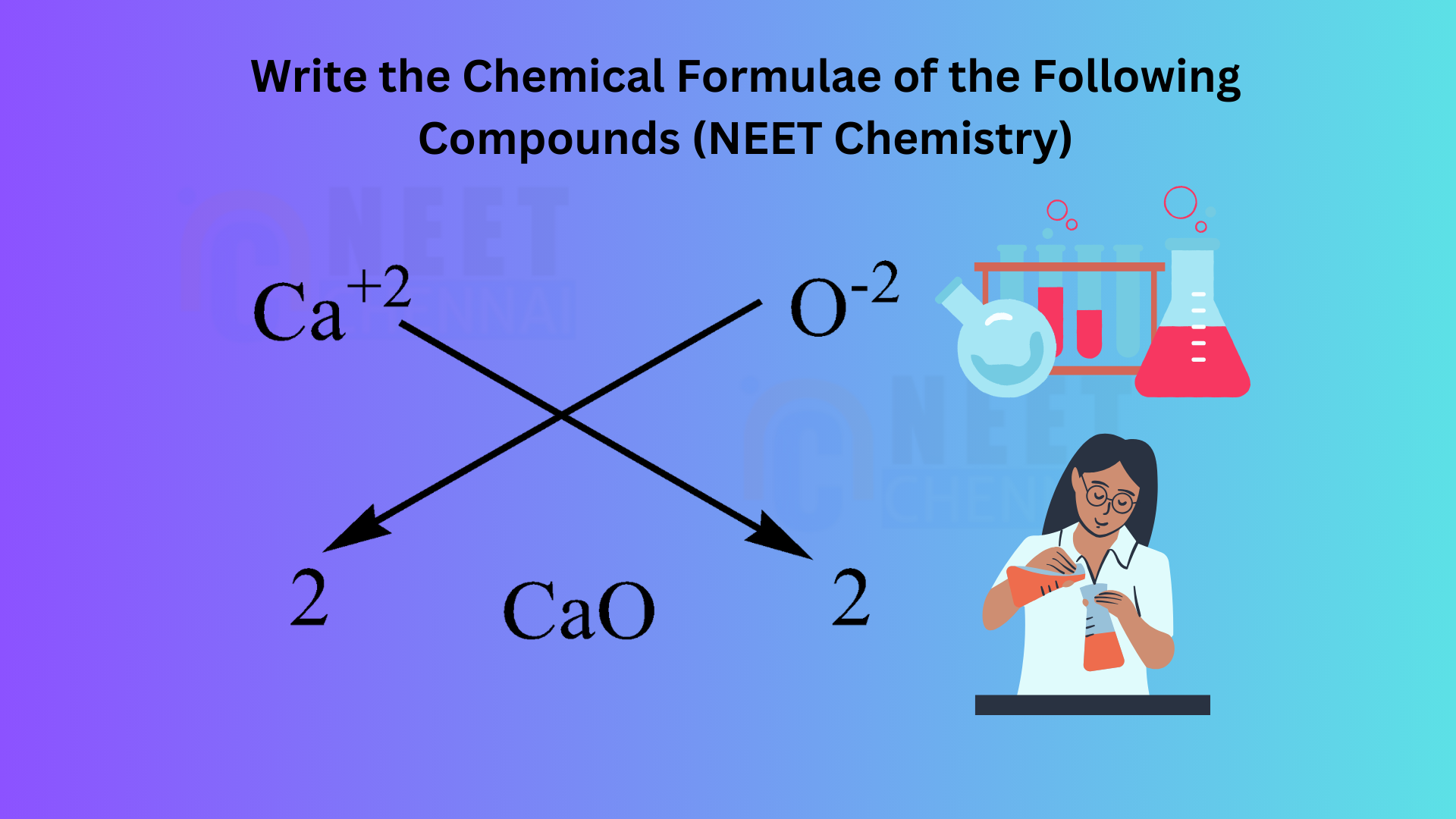
B.Calcium oxide
We have to know that the atomic number of calcium 2020 and has a valency of 2, it means it has 2 two atoms in the outermost shell for bonding. The atomic number of Oxygen is 88 and has a valency of 2, it has 6 atoms in the outermost shell, it needs more 2 to complete the octet. Hence, we need one calcium atom to bond with one oxygen atom.
We can use the criss-cross method for this compound as follows:
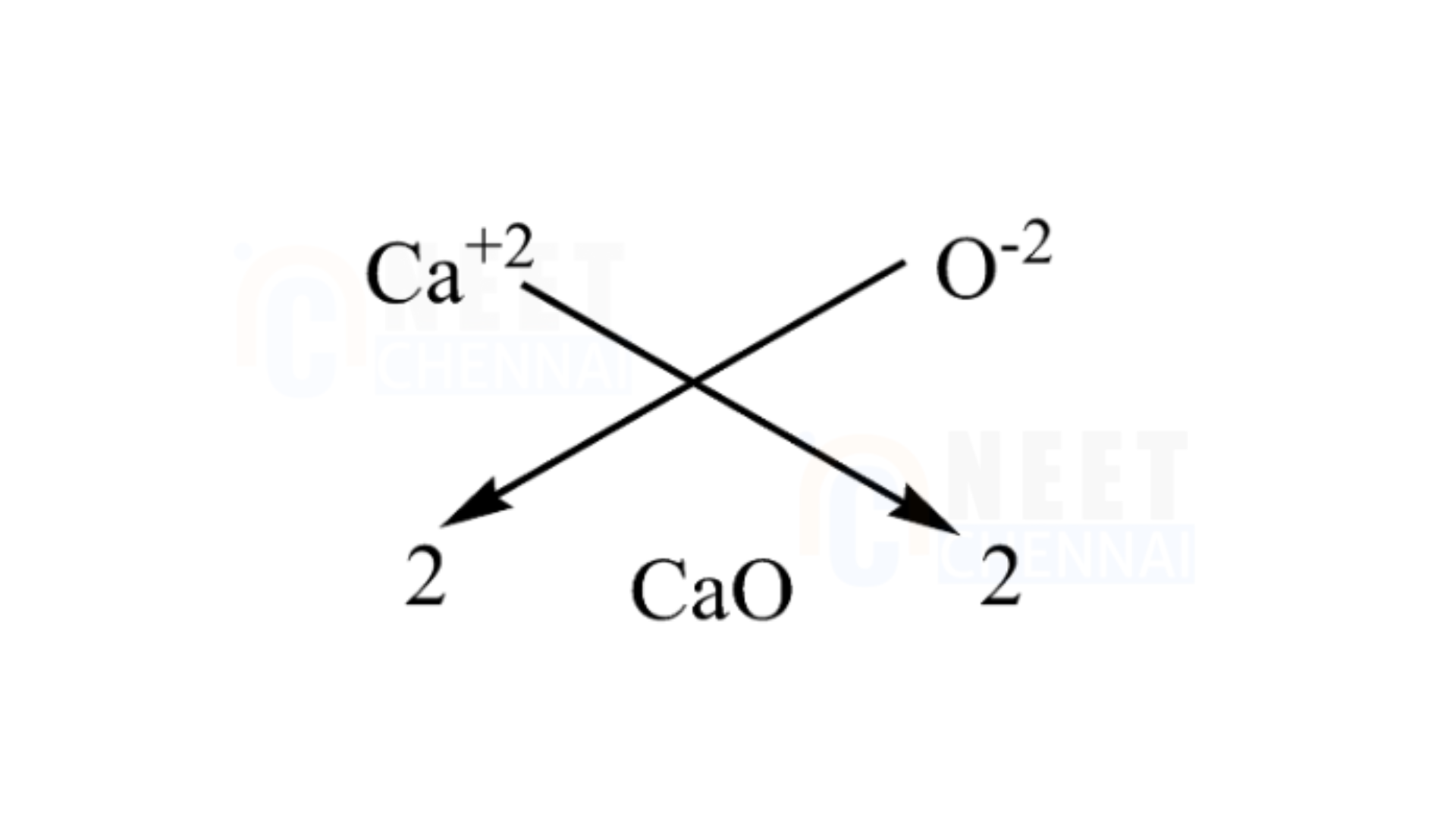 Thus, the chemical formula for magnesium chloride is CaO.
Thus, the chemical formula for magnesium chloride is CaO.
C. Copper nitrate
It is essential to note that copper has an atomic number of 29 and possesses two electrons in its outermost shell for bonding. In contrast, a nitrate molecule has only one valence electron. Therefore, we require two nitrate molecules to fulfill the valency of one copper atom.
We can use the criss-cross method for this compound as follows:
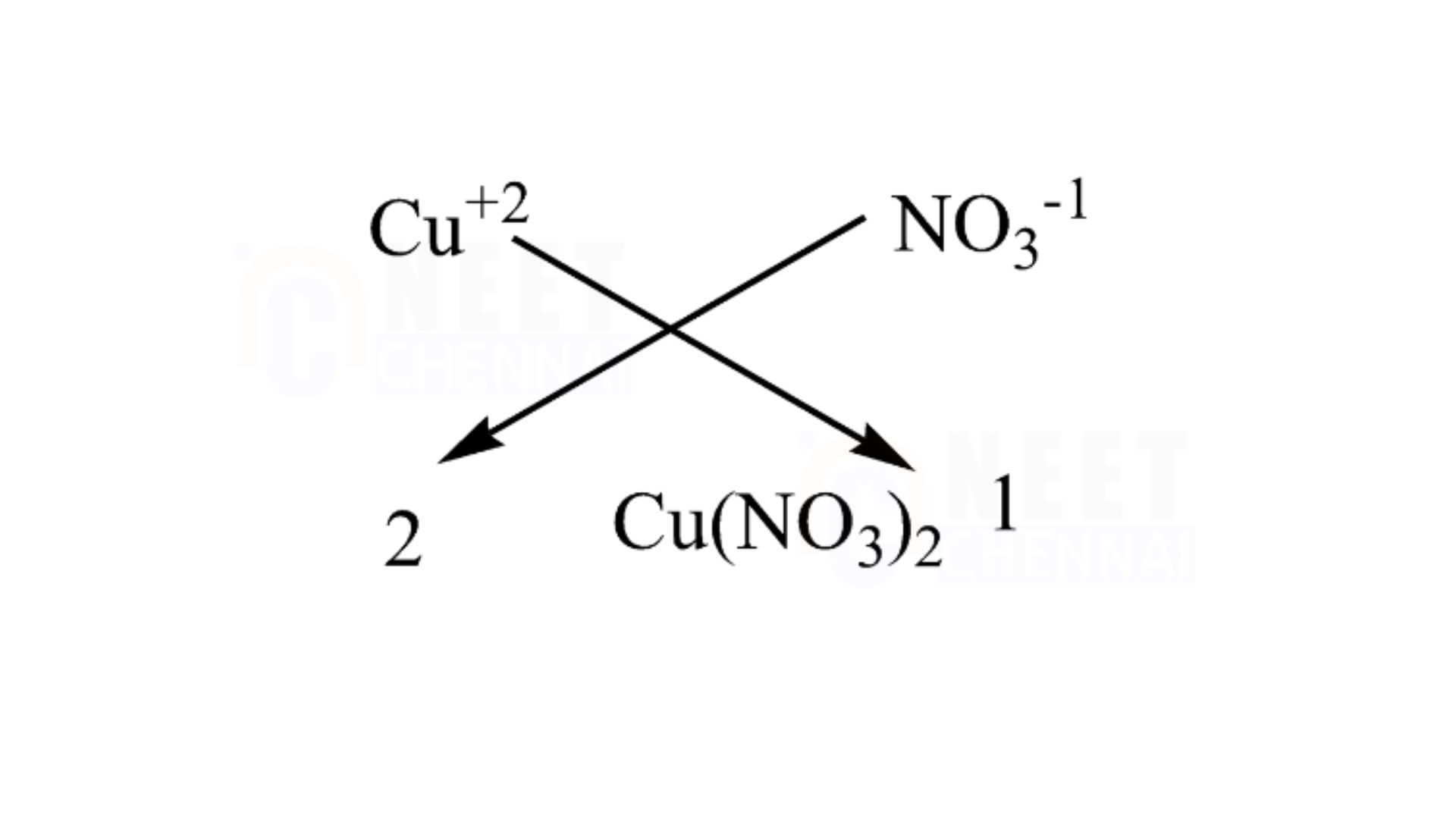 Therefore, the chemical formula of magnesium chloride is Cu(NO3)2.
Therefore, the chemical formula of magnesium chloride is Cu(NO3)2.
D. Aluminium chloride
It is important to recognize that aluminum has an atomic number of 13 and a valency of 3, while a chlorine atom has a valency of 1 due to having 7 electrons in its outermost shell. Therefore, we need three chlorine atoms to fulfill the valency of one aluminum atom.
We can use the criss-cross method for this compound as follows:
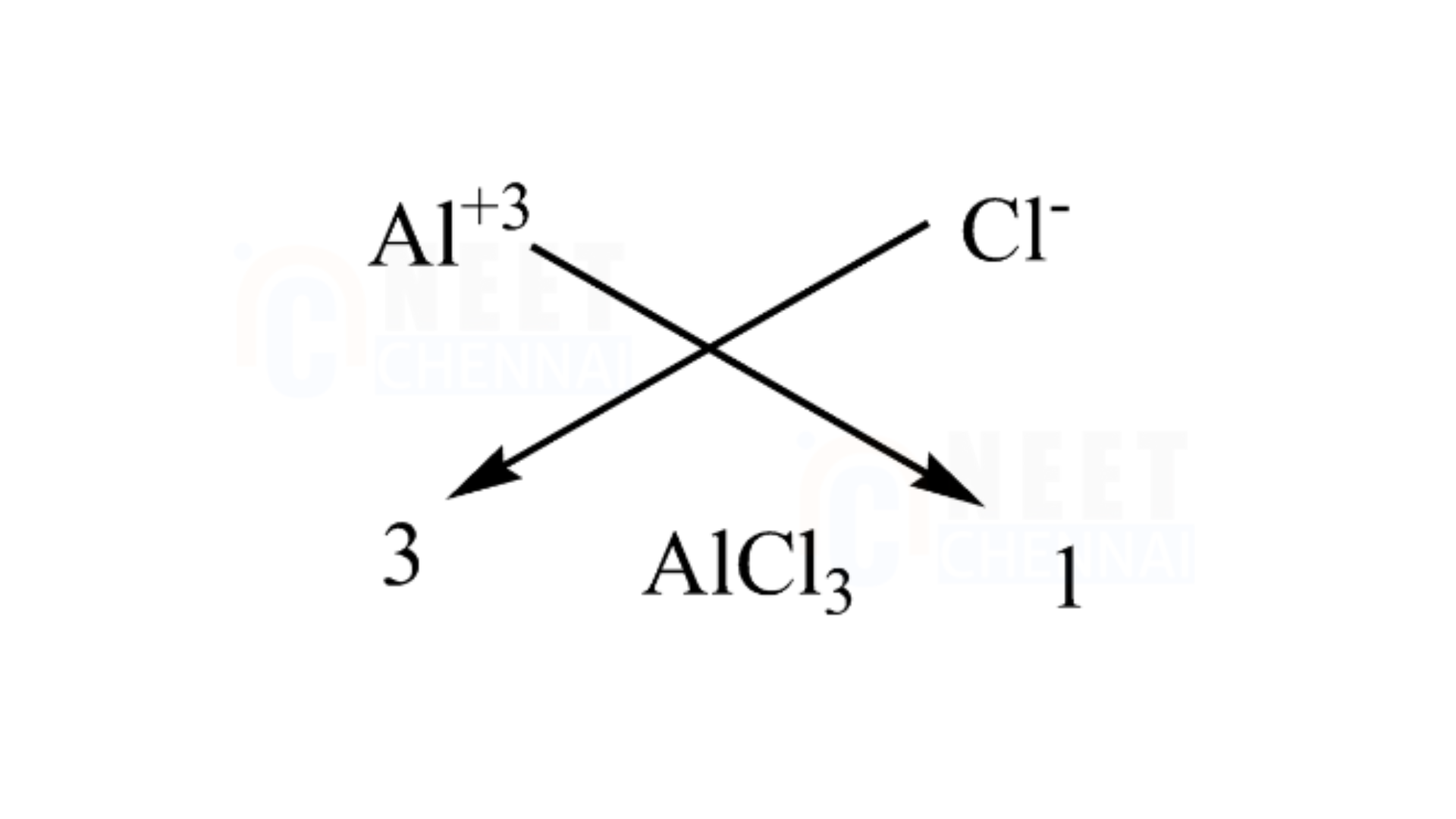 Therefore, the chemical formula of magnesium chloride is AlCl3.
Therefore, the chemical formula of magnesium chloride is AlCl3.
E. Potassium nitrate
It is essential to remember that potassium has an atomic number of 19 and a valency of 1, while nitrate also has a valency of 1, as it requires one additional atom to complete its octet. Therefore, only one molecule of nitrate is needed for one potassium atom.
We can utilize the criss-cross method for this compound as follows:
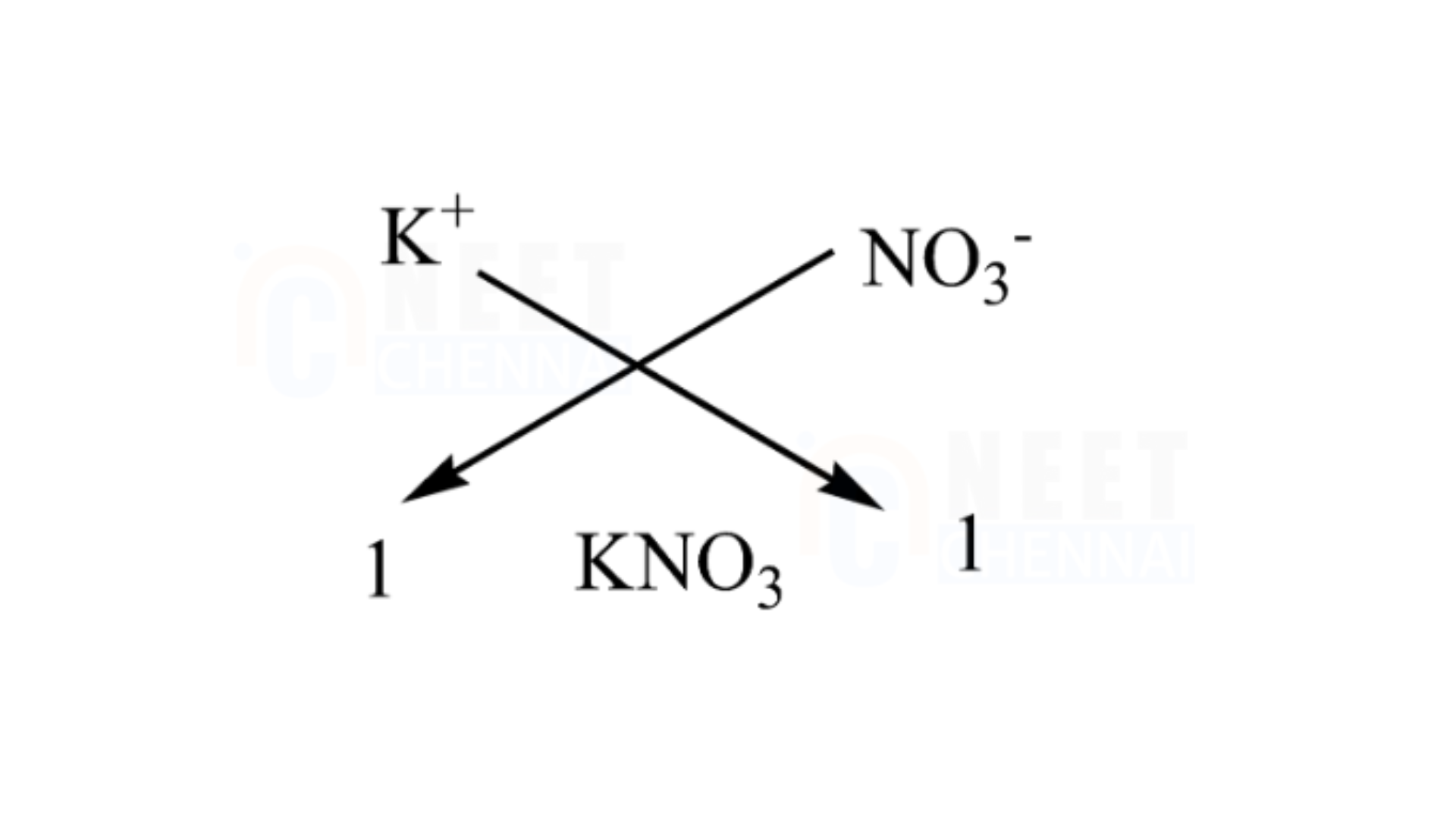 Thus, the chemical formula for magnesium chloride is KNO₃.
Thus, the chemical formula for magnesium chloride is KNO₃.
Note: As we know, the criss-cross method is the most effective way to determine the correct chemical formula of a compound. It is typically employed to derive the formula for the bonding between a metal and a non-metal to form ionic bonds. The charges of the two ions are ignored, and the value of the ions is crossed over to become the subscript for the corresponding atoms.