Hint: i) Transition elements involve the filling of d-orbitals, which have a smaller energy gap compared to their higher orbitals.
ii) Lanthanides are the elements in the sixth period beginning with lanthanum, which involve the filling of 4f orbitals, while actinides are the elements in the seventh period starting with actinium, which involve the filling of 5f orbitals.
Complete step by step answer:
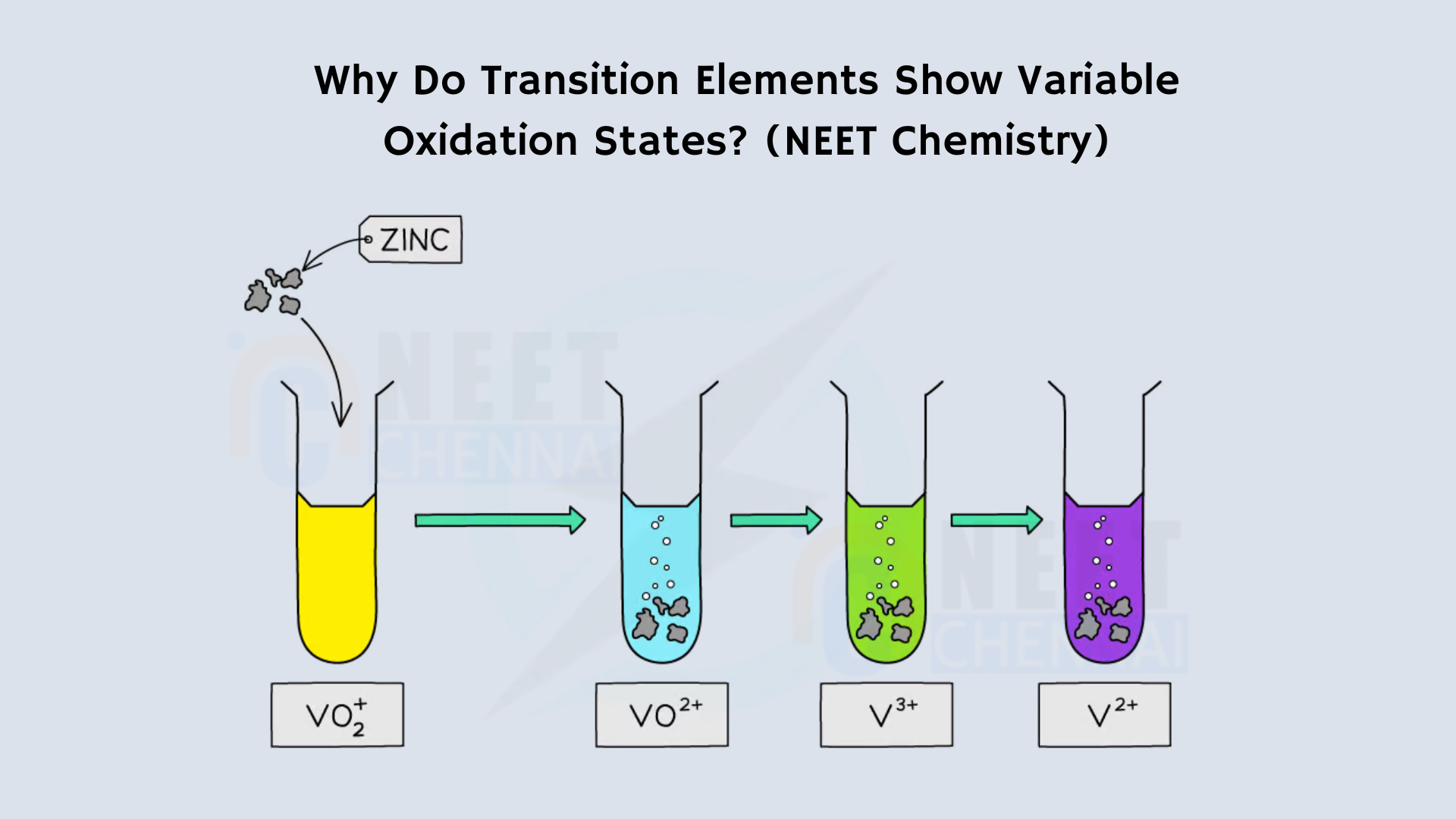
i) Transition metals are also referred to as d-block elements. They exhibit variable oxidation states because the (n-1)d orbitals are empty and energetically closer to the outermost ns orbital. These orbitals are never completely filled, allowing them to accommodate additional electrons in the (n-1)d orbitals.
For example, let’s examine the transition metal manganese (Mn). With an atomic number of 25, its electron configuration is:
\([\text{Ar}] 3d^5 4s^2\)It shows oxidation states from +2 to +7. The generally shown oxidation states are +2, +4 and +7.
To show +2 oxidation state it loses two electrons from the outer 4s orbital and to show +4 oxidation state four electrons are removed from the outer orbitals.
Moreover, these elements possess electrons in two distinct orbital states, namely ns and (n-1)d. The energy gap between these ns and (n-1)d orbitals is minimal, allowing both to share electrons during bond formation, which means that both can participate in bonding.
ii) Distinctions between lanthanides and actinides.
| Lanthanides |
Actinides |
| This term refers to the sixth period of the periodic table. | This is the name given to the seventh period in the periodic table. |
| It involves the filling of 4f and 6s orbital. | It involves filling of 5f and 7s orbital. |
| These show less tendency to form complex compounds. | These have more tendency to form complex compounds. |
| These form compounds which are less basic than actinide compounds. | These compounds tend to be more basic in nature. |
| Most of the lanthanides are non-radioactive (except promethium). | All the actinides are radioactive in nature. |
Note:
i) The most stable oxidation state for transition metals is typically +3. These metals form stable cations, allowing them to lose various electrons and exhibit a range of oxidation states.
ii) Lanthanides involve the filling of 4f orbitals, which is the first f-orbital. Therefore, this series is also referred to as the first transition series.