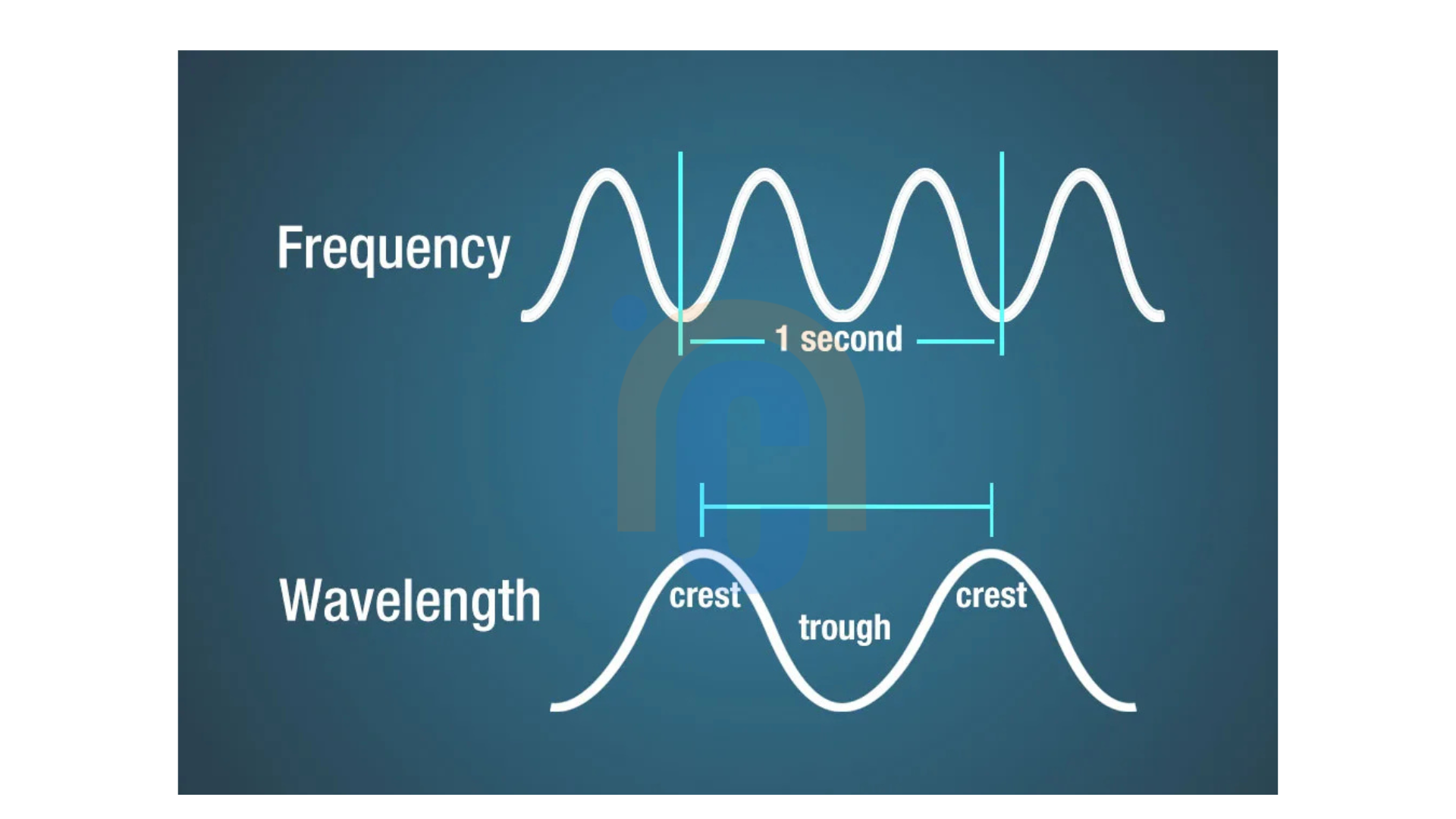
What is the relationship between frequency and wavelength of electromagnetic radiation?
Hint: Energy, wavelength, and frequency can all be used to characterize the electromagnetic spectrum. Each way of understanding the electromagnetic spectrum is mathematically connected to the others. The relationships between energy, wavelength, and frequency can be stated as wavelength equals the speed of light divided by the frequency.
Complete answer:
Light’s wavelength and frequency are inextricably linked. The shorter the wavelength, the higher the frequency. Therefore, frequency and wavelength are inversely related. Since all light waves travel at the same speed in a vacuum, the number of wave crests passing a specific point in one second is influenced by the wavelength. For a wave with a short wavelength, this count—referred to as the frequency—will be greater than that of a wave with a long wavelength. The relationship between the wavelength and frequency of electromagnetic waves is expressed by the equation λν = c, where λ represents the wavelength, ν denotes the frequency, and c is the speed of light.
Explanation:
The wavelength and frequency are inversely proportional.
c=ν⋅λ
(The speed of light is directly proportional to ν and λ )
\(\frac{c}{ν}=λ\)(The speed of light is directly proportional to ν , and ν is inversely proportional to λ
\(\frac{c}{λ}=ν\) (The speed of light is directly proportional to λ and λ is inversely proportional to νNote: Spectroscopy is the precise study of colour, broadened from visible light to all bands of the electromagnetic spectrum; indeed, spectroscopy began as a study of the wavelength dependence of visible light scattered by a prism absorption by gas phase materials. For spectroscopy, nearly all frequencies and wavelengths of electromagnetic radiation can be employed.