The principle involved in paper chromatography is:
A) Adsorption
B) Partition
C) Solubility
D) Volatility
Hint: Chromatography is an analytical method used to separate mixtures into their individual components, with each component moving at a unique speed. Various chromatographic techniques exist, such as column chromatography, thin-layer chromatography, gas chromatography, and paper chromatography.
Complete step-by-step answer:
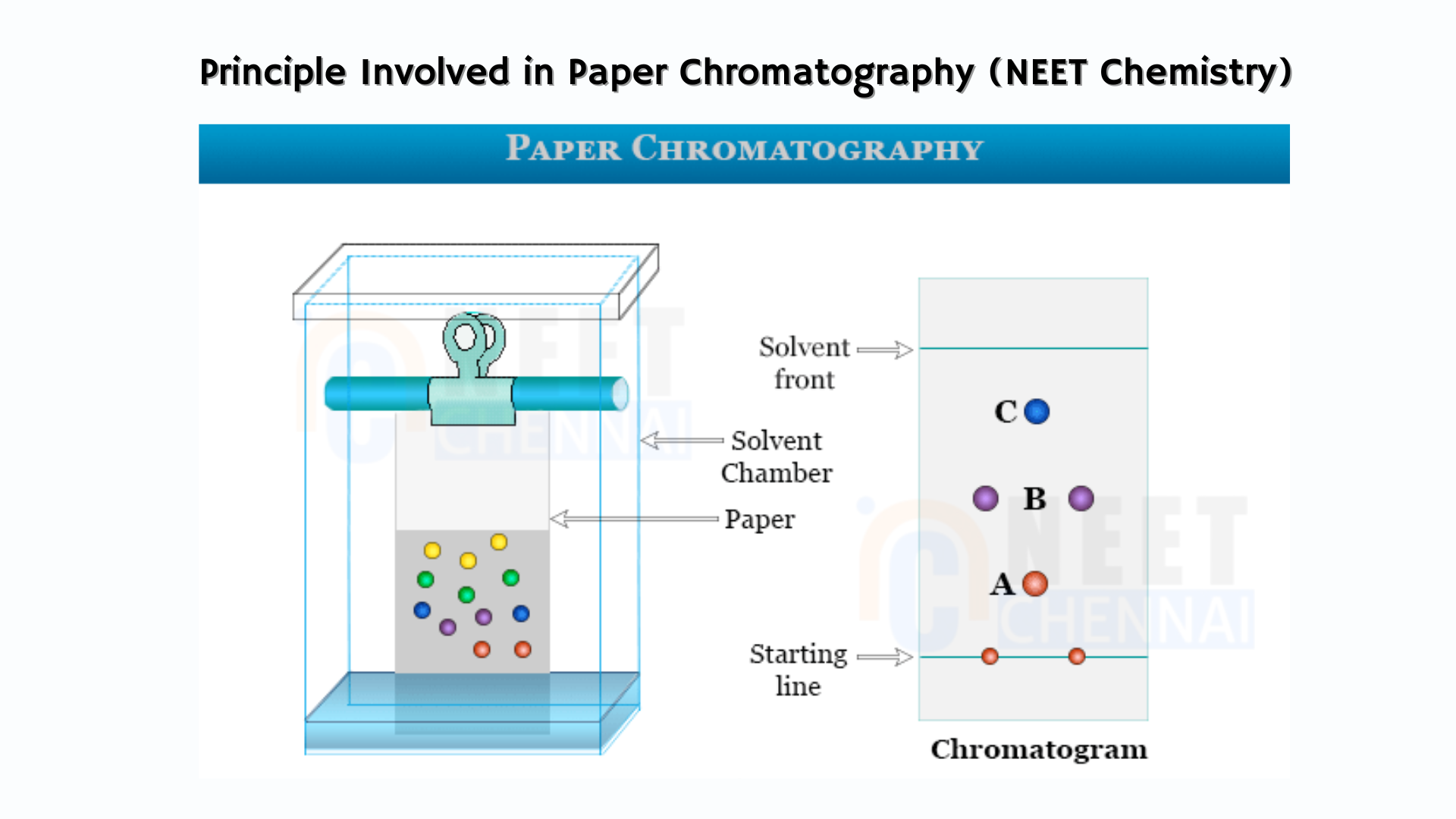
Paper chromatography is a chromatographic technique used to separate a mixture of colored components on paper, where each component travels at a different speed, resulting in separation. Components with higher speeds move faster than those with lower speeds.
The principle behind paper chromatography is partition. In this technique, two phases are involved: the stationary phase and the mobile phase. Here, water trapped in the paper serves as the stationary phase, while the solvent acts as the mobile phase. As the solvent travels, the mixture’s components move at varying speeds, causing them to separate based on their distribution between the mobile and stationary phases.
Therefore, paper chromatography operates on the partition principle. Options (A) adsorption, (C) solubility, and (D) volatility are incorrect. Thus, the correct answer is option (B).
Note:
i) Adsorption is a process where molecules of one substance adhere to the surface of another.
ii) Partition refers to the separation or distribution of a substance into distinct areas.
iii) Solubility is a substance’s capacity to dissolve in a solvent.
iv) Volatility is a substance’s tendency to transform into vapor.