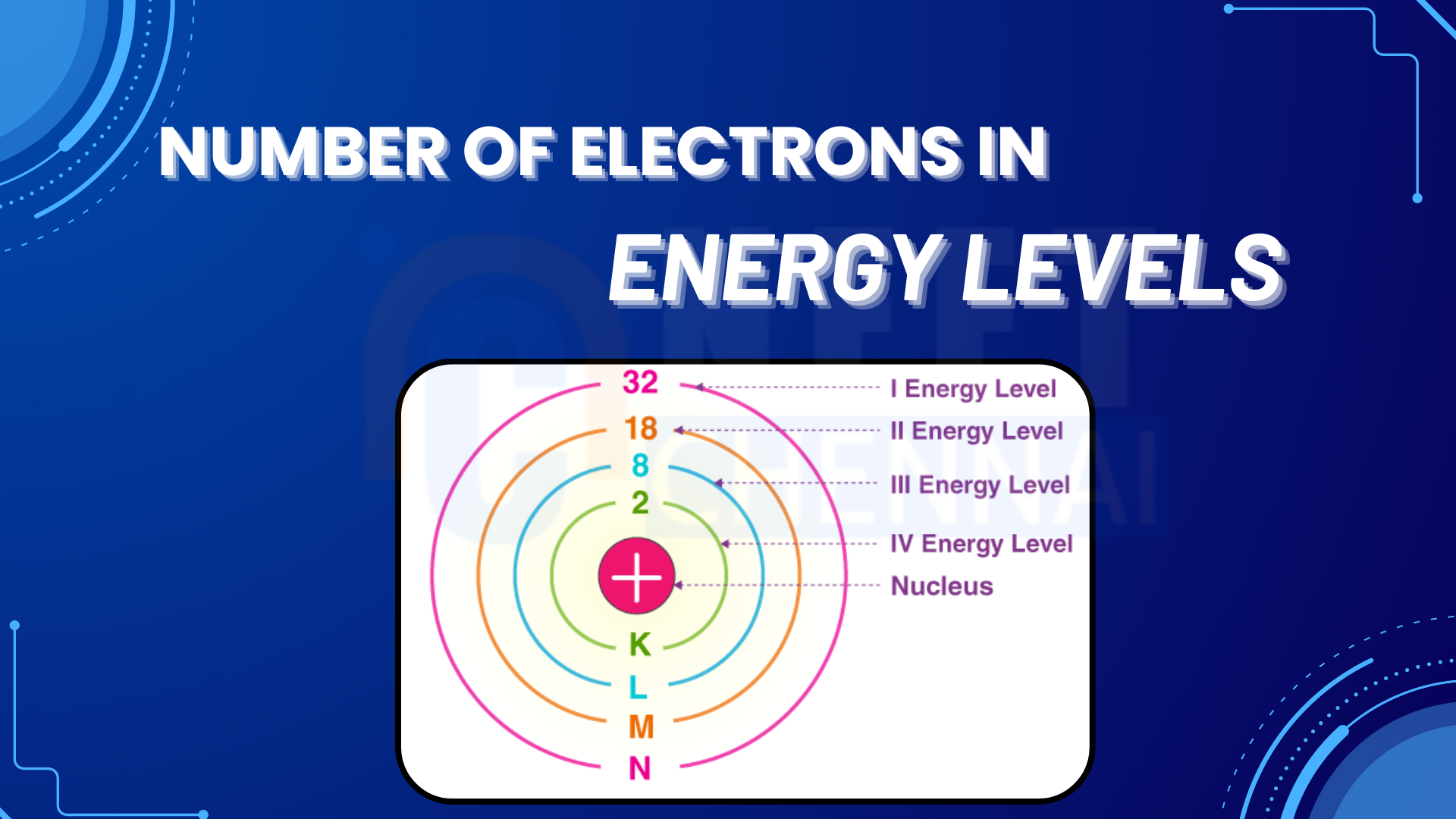
In general , the number of electrons in the outermost shell of a metal atom is :
A.1
B.1-3
C.1-8
D.8
Hint: All the elements in the periodic table exist in their most stable forms. The elements in the periodic table either give up or take in electrons to achieve stability.
Complete step by step answer:
Some elements achieve stability by completing a duplet, meaning they become stable when they have two electrons in their outermost shell, as their 1s orbital can only hold a maximum of two electrons. Hydrogen and Helium are the two elements that stabilize by completing their duplet.
All other elements achieve stability by completing their octet, meaning they have eight electrons in their outermost shell, as most atoms’ valence shells can accommodate a maximum of eight electrons.
Elements are classified into two categories based on their ability to give or accept electrons: metals and non-metals.
Metals are elements that donate electrons, while non-metals are those that accept electrons. Metals donate electrons to complete their octet and achieve a noble gas configuration. They typically have one, two, or three electrons in their valence shell, so it is easier for them to lose electrons rather than gain more.
Non-metals, on the other hand, accept electrons to complete their octet because they have five, six, or seven electrons in their valence shell, making it easier for them to gain electrons rather than lose them.
When metals donate electrons, they form cations, which are positively charged ions, depending on the number of electrons they donate. For instance, sodium donates one electron, forming a +1 charge. The number of electrons donated determines the valency of the metal atom.
Therefore, the right answer is Option B.
Note:
Metals are found on the left side of the periodic table and make up 78 percent of all the elements present. They are excellent conductors of both heat and electricity.