Write the mechanism of hydration of ethene to yield ethanol.
Hint:In a hydration reaction, a water molecule interacts with an unsaturated compound rather than a saturated one, resulting in the formation of an alcohol.
Complete step-by-step solution:
Let us see the example of hydration reaction:
In this process, ethene molecules react with sulfuric acid to produce ethanol, an alcohol. During a hydration reaction, water interacts with an alkene to form an alcohol. Ethanol can be synthesized through the reaction of an alkene with water. Ethene reacts with sulfuric acid, leading to the formation of ethanol.
The transformation of ethene into ethanol takes place in three distinct stages, which are as follows:
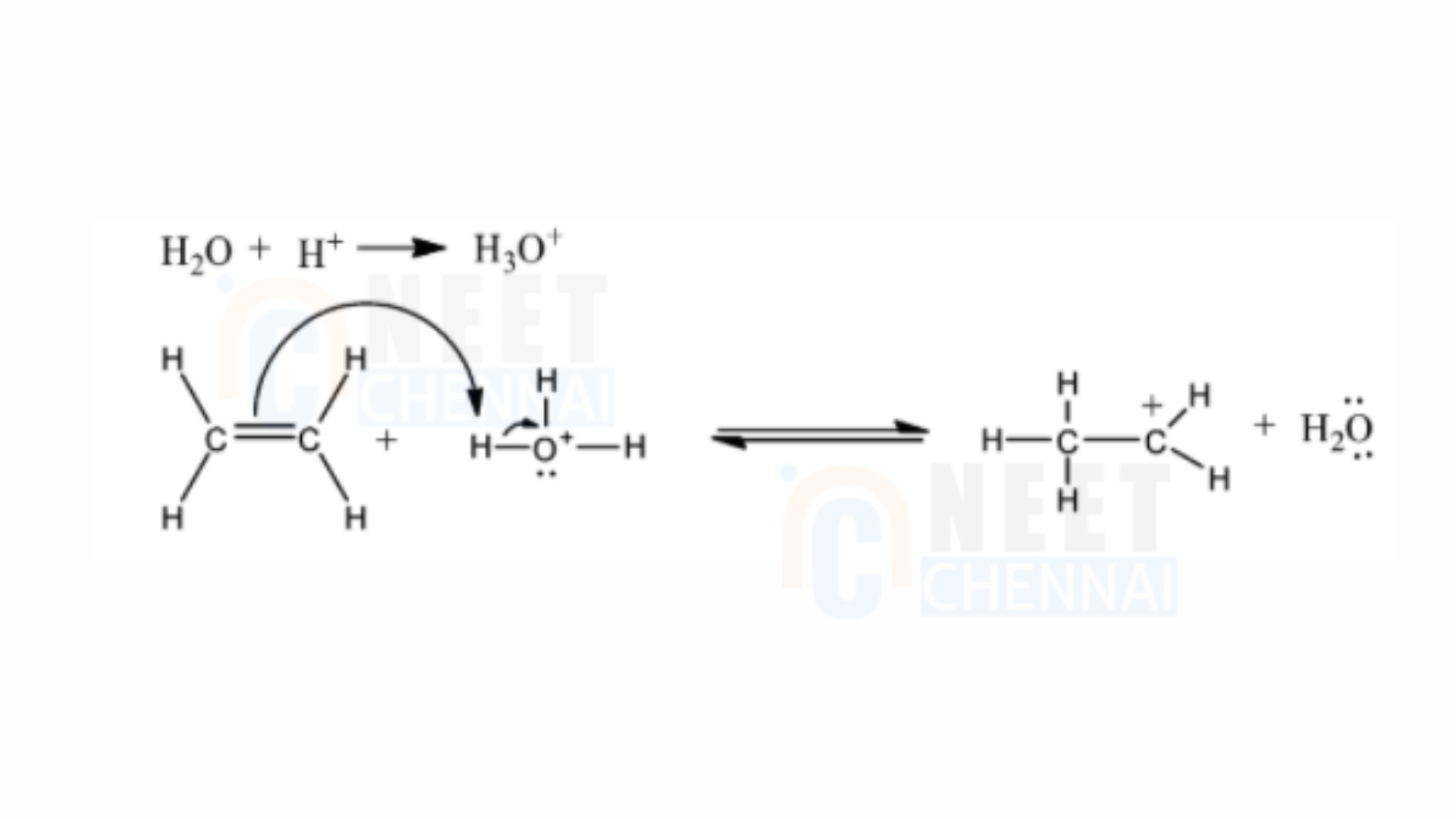
1) An electrophilic interaction with the hydronium ion occurs, leading to the protonation of ethene and resulting in the formation of a carbocation.
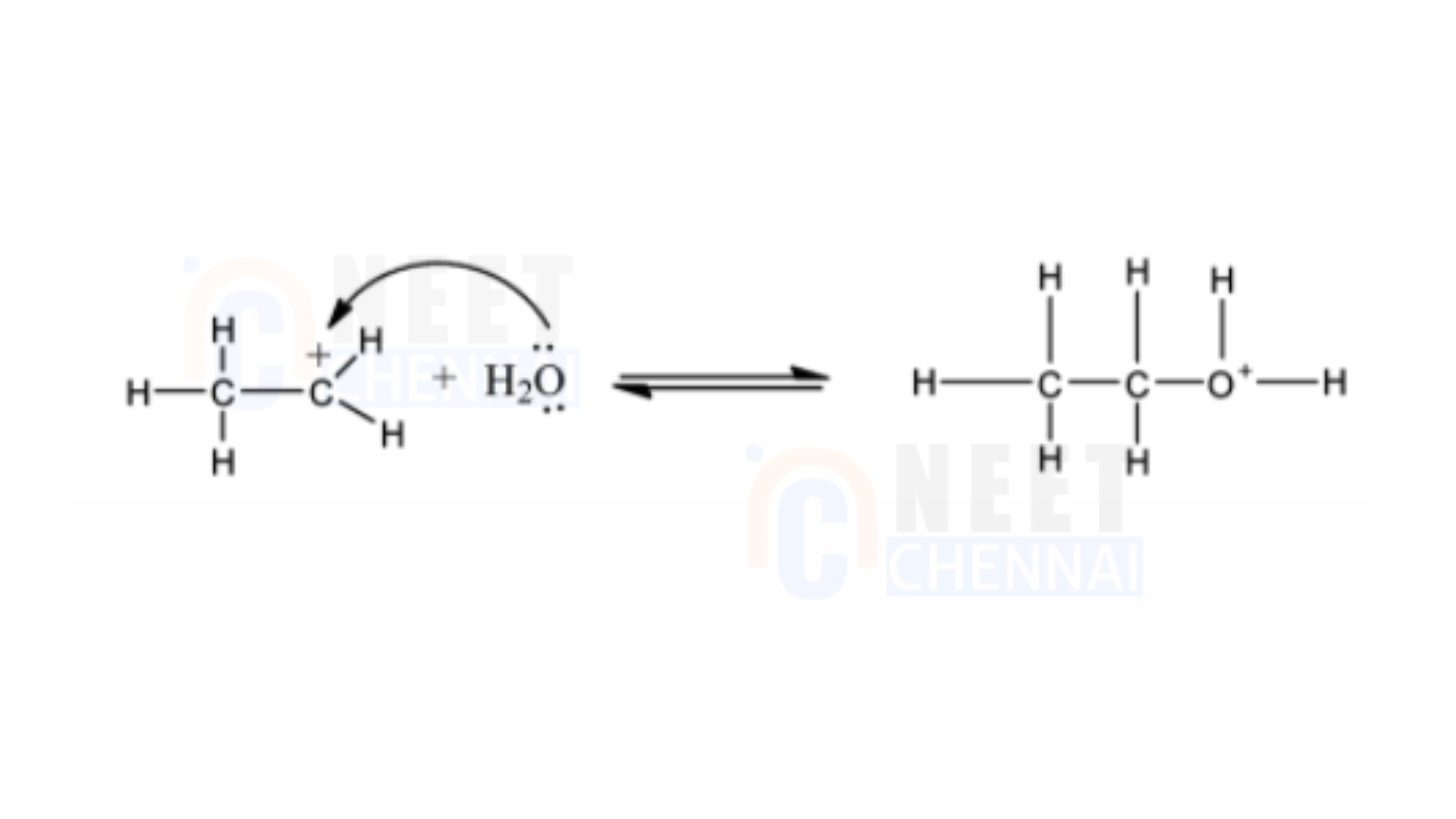
 2) In the subsequent step, the water molecule interacts with and attaches to the carbocation.
2) In the subsequent step, the water molecule interacts with and attaches to the carbocation.
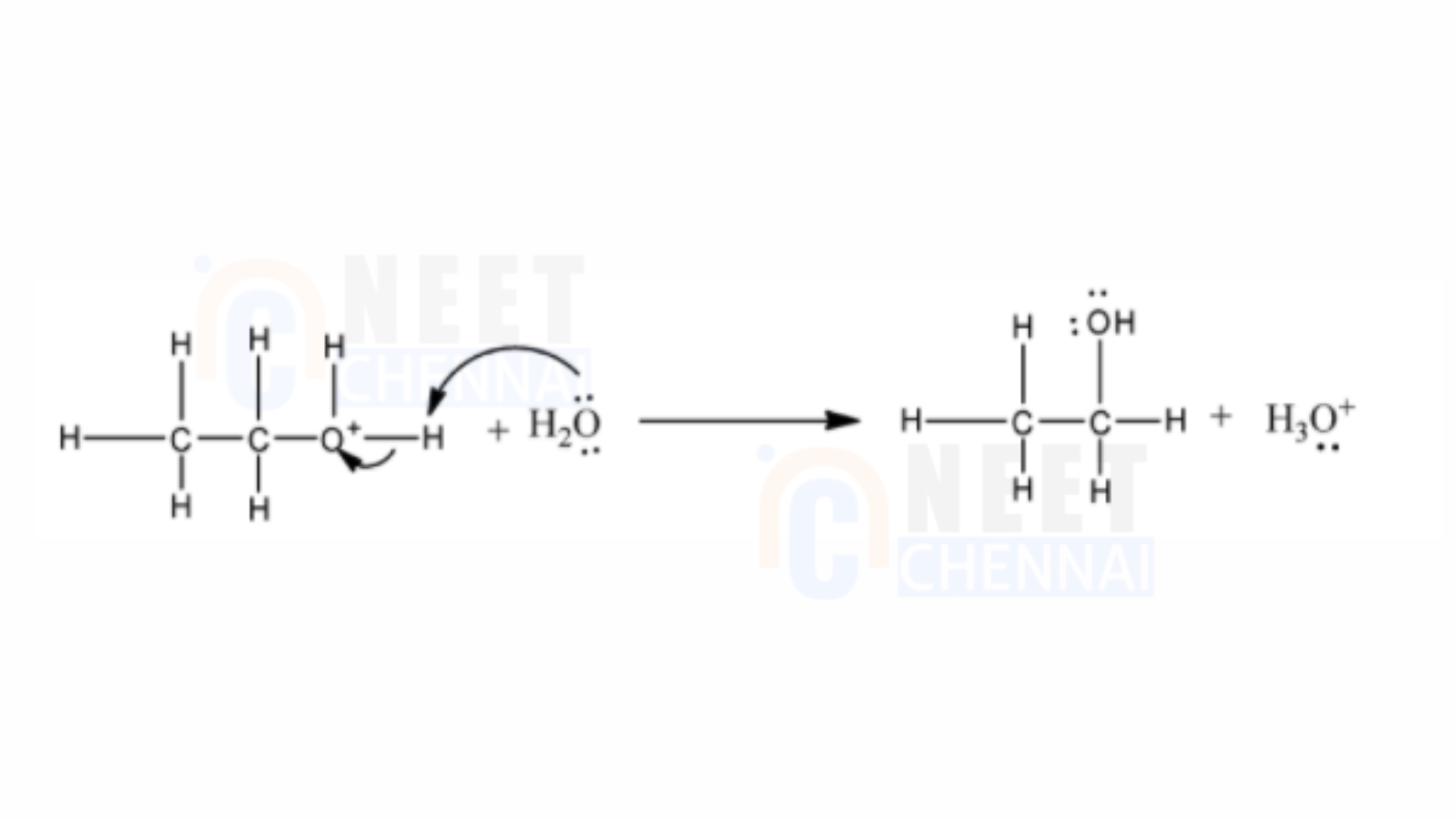
 3) Finally, the removal of a proton leads to the formation of ethanol.
3) Finally, the removal of a proton leads to the formation of ethanol.
 Note: Students might find it challenging to differentiate between a hydration reaction and a hydrolysis reaction. In a hydration reaction, water interacts with an unsaturated compound to produce an alcohol. However, in a hydrolysis reaction, the addition of water causes the breaking of chemical bonds.
Note: Students might find it challenging to differentiate between a hydration reaction and a hydrolysis reaction. In a hydration reaction, water interacts with an unsaturated compound to produce an alcohol. However, in a hydrolysis reaction, the addition of water causes the breaking of chemical bonds.