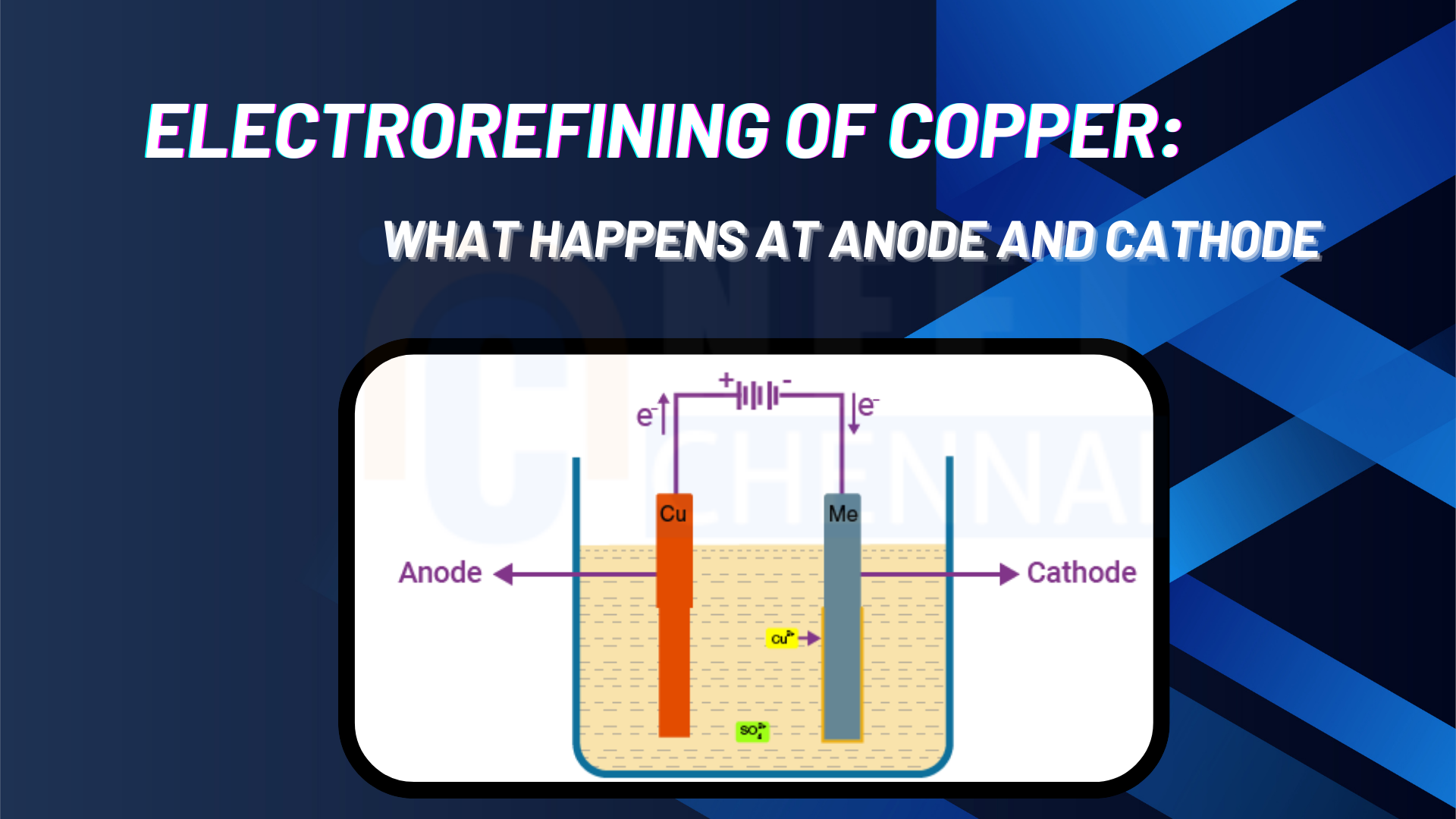
For the electro-refining of copper.
(i) What is the cathode made up of?
(ii) Write the reaction that takes place at the anode.
Hint:
(i) In the process of electrolytic refining of copper, both electrodes are composed of copper metal. One electrode is made of pure copper, while the other consists of impure copper.
(ii) It is well known that oxidation takes place at the anode and reduction happens at the cathode.
Complete Step by step answer:
(i) In the process of electrolytic refining of copper, the anode is the positive electrode made of impure copper blocks, while the electrolyte consists of a solution of copper sulfate and sulfuric acid. The cathode, which is the negative electrode, is composed of pure copper. As a result, the cathode is made up of pure copper.
(ii) During electrolysis, copper sulfate dissociates to release copper ions (Cu²⁺) and sulfate ions (SO₄²⁻). At the cathode, the copper ions are reduced by gaining electrons. The deposition of concentrated copper metal onto the cathode is facilitated by coating the cathode with graphite. The impurities present in the impure copper at the anode react with sulfate ions to form metal sulfates, which then dissolve in the electrolyte solution. Certain impurities, such as silver and gold, do not dissolve and settle at the bottom as anode mud (sludge or residue). The reaction occurring at the anode involves the oxidation of copper metal into copper ions.
Cu → Cu2++2e−
Note: Blister copper has a purity of approximately 98 to 99 percent. The purity of copper can be increased to 99.95 percent through the electro-refining process. This high-purity copper can then be utilized in electrical components.