Differentiate between exergonic and endergonic reactions.
Hint: An exergonic reaction is a chemical reaction that releases free energy, while an endergonic reaction is a chemical reaction in which energy is absorbed.
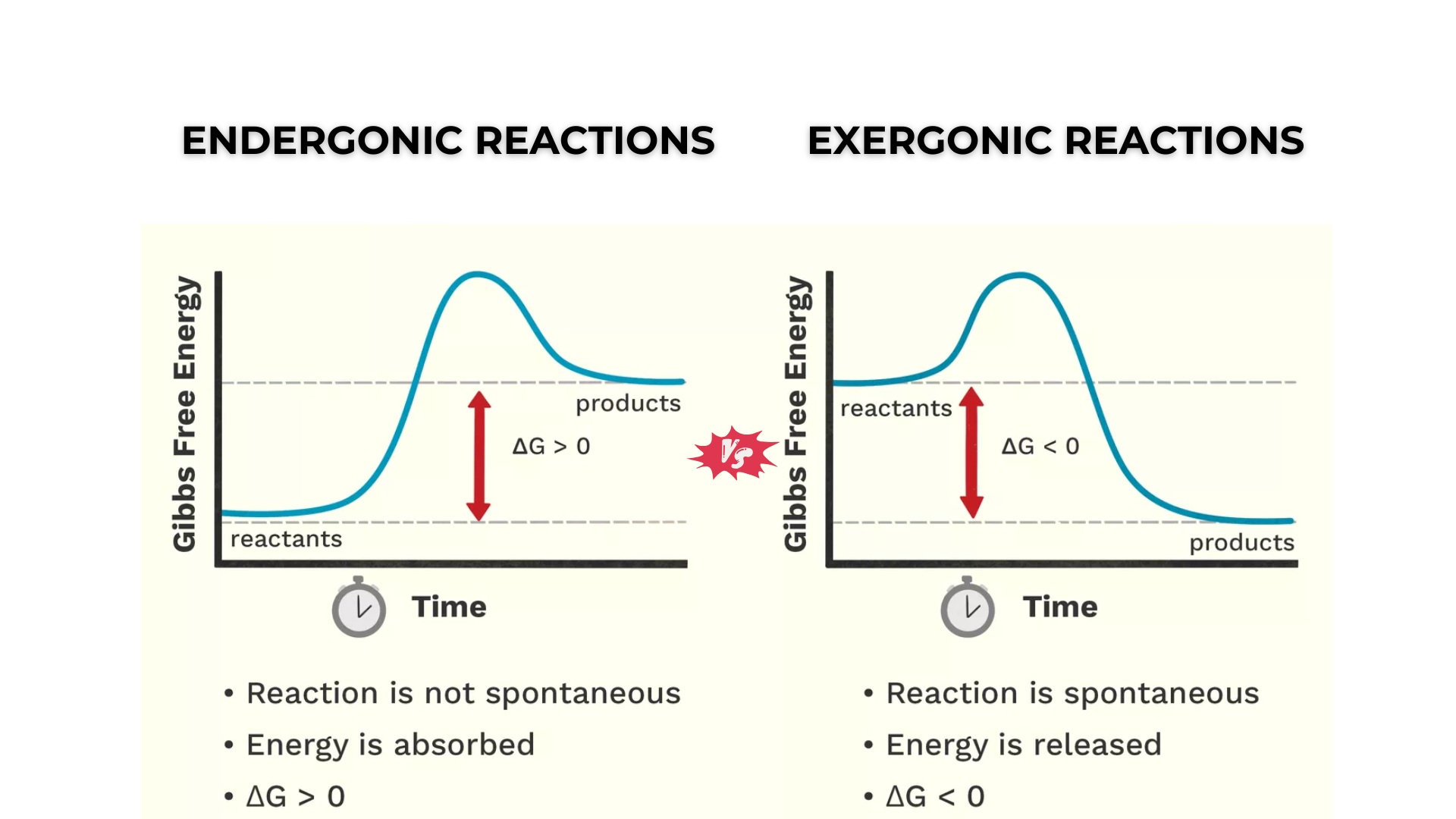
Complete answer: An exergonic reaction is a type of spontaneous reaction in which free energy is released, resulting in negative free energy (less than zero).
In contrast, endergonic reactions are those in which energy is absorbed into the system, resulting in a positive free energy (greater than zero).
Free energy, also known as Gibbs free energy (∆G), represents the total usable energy. When free energy is negative (-∆G), it shows that energy is being released, while if it is positive, it indicates that energy is being taken in or stored.
|
Exergonic reaction |
Endergonic reaction |
| An exergonic reaction is a type of reaction where free energy is released. | Endergonic reactions are reactions in which free energy is absorbed. |
| In this case, Gibbs free energy is negative. | In this case, Gibbs free energy is positive. |
| Exergonic reactions signify that energy is being released into the system. | Endergonic reactions signify that energy is being absorbed by the system. |
| All exothermic reactions are exergonic. | All endothermic reactions are endergonic. |
| Exergonic reactions do not require energy to initiate. | Endergonic reactions always require energy to start. |
| It is a downhill process. | It is an uphill process. |
| Examples include fatty acid catabolism, glycolysis, and cellular respiration. | Examples include the formation of DNA/RNA, protein formation, and the synthesis of fatty acids. |
Note:
Endergonic and exergonic reactions serve as the foundation for the processes occurring in different organisms.
Catalysts may be needed for certain reactions to occur at a noticeable rate.