What is Shell and Orbit: Introduction
Atoms are the basic building blocks of matter, and for a long time, scientists believed they were indivisible. However, further investigation into subatomic particles revealed that atoms can be broken down into smaller parts. These subatomic particles include electrons, protons, and neutrons, with protons and neutrons forming the nucleus at the core of the atom.
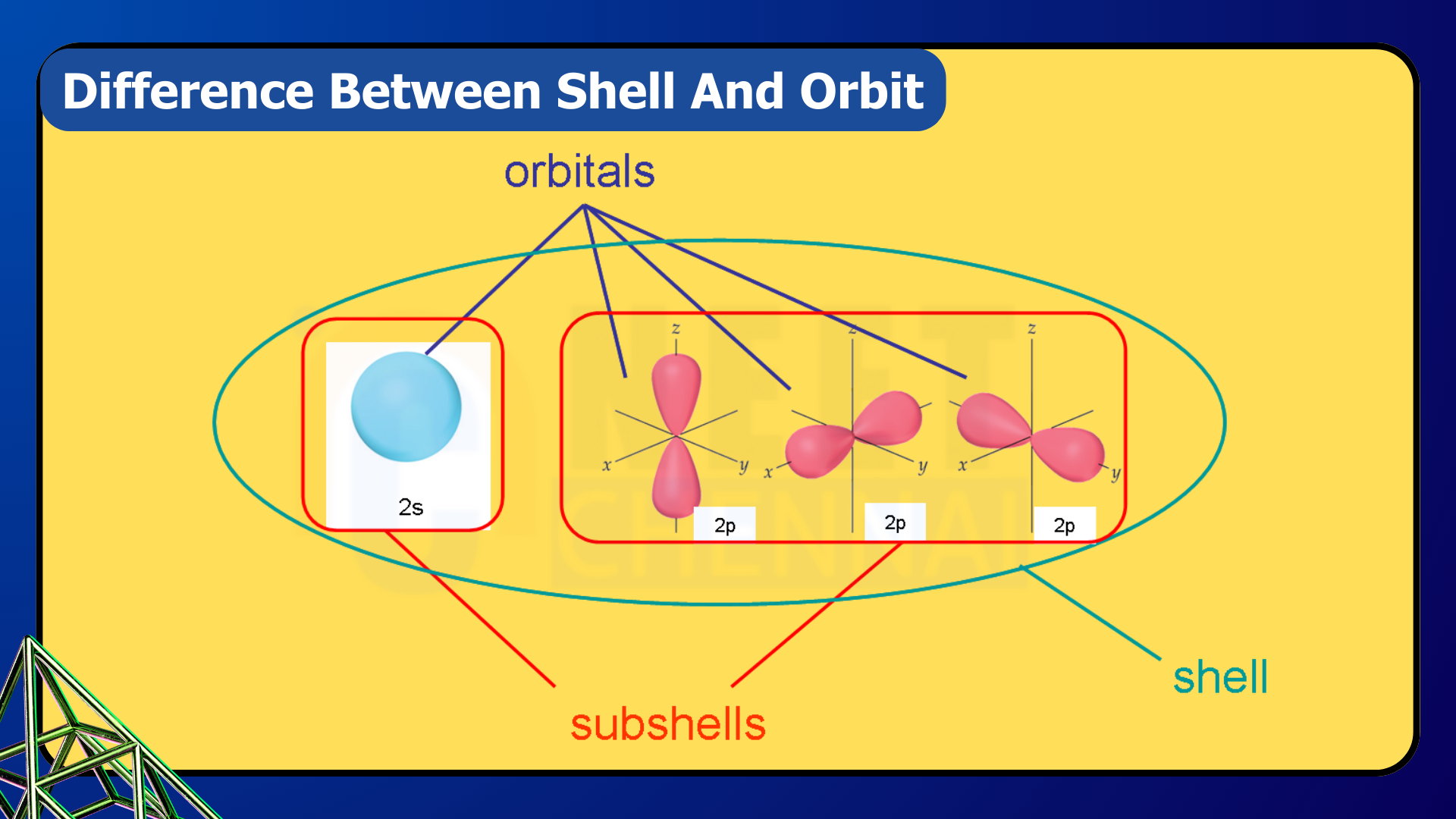
Electrons, in contrast, move around the nucleus in specific pathways. Although the precise location of an electron cannot be exactly determined, it travels along likely routes known as shells, subshells, and orbitals. Shells and subshells consist of electrons that share the same principal quantum number and angular momentum quantum number, respectively. Orbitals, on the other hand, hold electrons with opposite spins but at the same energy level. This distinction marks the primary difference between shells, subshells, and orbitals.
Shell in Atomic Structure:
Shell: Electrons surrounding an atom’s nucleus move along pathways called shells, also known as energy levels. These shells are organized based on the energy of the electrons they contain, with the shell having the lowest energy positioned nearest to the nucleus. Each subsequent energy level is situated farther from the previous one. These shells are labeled using letters like K, L, M, N, etc. The shell with the lowest energy is referred to as the K shell. However, scientists use quantum numbers to identify these shells.
Quantum Number:
Each shell has a distinct quantum number, known as the principal quantum number. The shell with the lowest energy level is assigned n=1. It’s important to understand that not all shells have the same electron capacity. The lowest energy shell can hold a maximum of 2 electrons, while the next level can accommodate up to 8 electrons. The highest number of electrons any shell can contain is 32, and no shell can surpass this limit. Higher energy levels can hold more electrons compared to lower ones. The presence of these shells signifies that an atom’s energy is quantized, meaning electrons around the nucleus possess discrete energy levels. Electrons within these shells can jump from one shell to another by either absorbing or releasing energy, but such transitions occur only if the energy absorbed or emitted equals the energy difference between the shells. If the energy is not an exact match, the transition cannot happen.
Orbit of an Atom:
Orbit: An orbit refers to the specific path or region where an electron moves around the nucleus of an atom. Simply put, an orbital describes the probable motion of an electron. A subshell, on the other hand, is made up of several orbitals. The number of orbitals in a subshell depends on the particular subshell, making the number of orbitals a unique feature of each subshell. However, each orbital can hold a maximum of two electrons. These electrons share the same energy level but have opposite spins, always paired with different spin orientations. According to Hund’s Rule, when electrons fill orbitals within a subshell, each orbital is occupied by a single electron before any are doubly occupied.
Let’s explore the concepts of shell and orbit through real-life examples.
Example 1: Atomic Structure
Take the element carbon (C) as an example. A carbon atom consists of a nucleus made up of protons and neutrons, with electrons occupying various energy levels, or shells, around the nucleus. The first shell (n = 1) can accommodate up to 2 electrons, while the second shell (n = 2) can hold a maximum of 8 electrons. In the case of carbon, the first shell contains 2 electrons, and the second shell holds 4 electrons, giving a total of 6 electrons.
Example 2: Celestial Bodies
Now, consider Earth’s orbit around the Sun. Earth moves in an elliptical orbit with the Sun positioned at one of the foci of the ellipse. This motion is controlled by the gravitational force between Earth and the Sun. Earth’s orbital path determines its location relative to the Sun, influencing the seasons, climate, and other astronomical events.
These real-world examples illustrate the practical relevance of shells and orbits in both atomic structures and celestial mechanics. Shells dictate the electron arrangement and chemical characteristics of atoms, while orbits define the motion and spatial relationships of planets and other celestial objects..
Difference Between Shell and Orbit
Sr. No. |
Category |
Shell |
Orbit |
1 |
Definition |
A specific energy level, or electron cloud, surrounds the atomic nucleus. |
This region indicates the most likely location for an electron within a shell. |
2 |
Representation |
Shells are denoted by principal quantum numbers (n). |
Orbitals are defined by a combination of principal (n), azimuthal (l), magnetic (m), and spin (s) quantum numbers. |
3 |
Energy Levels |
Each shell possesses a unique energy level, with the innermost shell having the lowest energy and the outermost shell having the highest. |
All orbitals within a shell share the same energy level. |
4 |
Maximum Electron Capacity |
The maximum electron capacity of a shell is calculated using the formula 2n², where n is the principal quantum number. |
Each orbital can accommodate a maximum of two electrons with opposing spins. |
5 |
Arrangement |
Shells are filled in order of increasing energy, with electrons occupying lower energy shells before moving to higher ones. |
Orbitals within a shell are organized by energy and filled according to the Aufbau principle, Hund’s rule, and the Pauli exclusion principle. |
6 |
Spatial Distribution |
Shells are depicted as concentric spherical layers surrounding the nucleus. |
Orbitals vary in shape (spherical, dumbbell, etc.) and orientation (along x, y, and z axes) within a shell. |
7 |
Relationship |
Each shell can contain one or more orbitals. |
Orbitals are subsets of shells, with each representing different energy states for the electron within that shell. |
Summary
In atomic structure, shells and orbitals are essential for understanding how electrons are distributed and behave within an atom. Shells represent the energy levels or electron clouds that surround the nucleus, while orbitals define the most likely regions where electrons can be located within each shell. Shells are identified by principal quantum numbers, with each shell capable of holding a specific number of electrons. Orbitals, however, describe the regions or pathways along which electrons move. The arrangement of electrons within shells and orbitals influences the chemical properties and reactivity of atoms. Together, shells and orbitals provide a framework for explaining atomic structure and electron configuration.