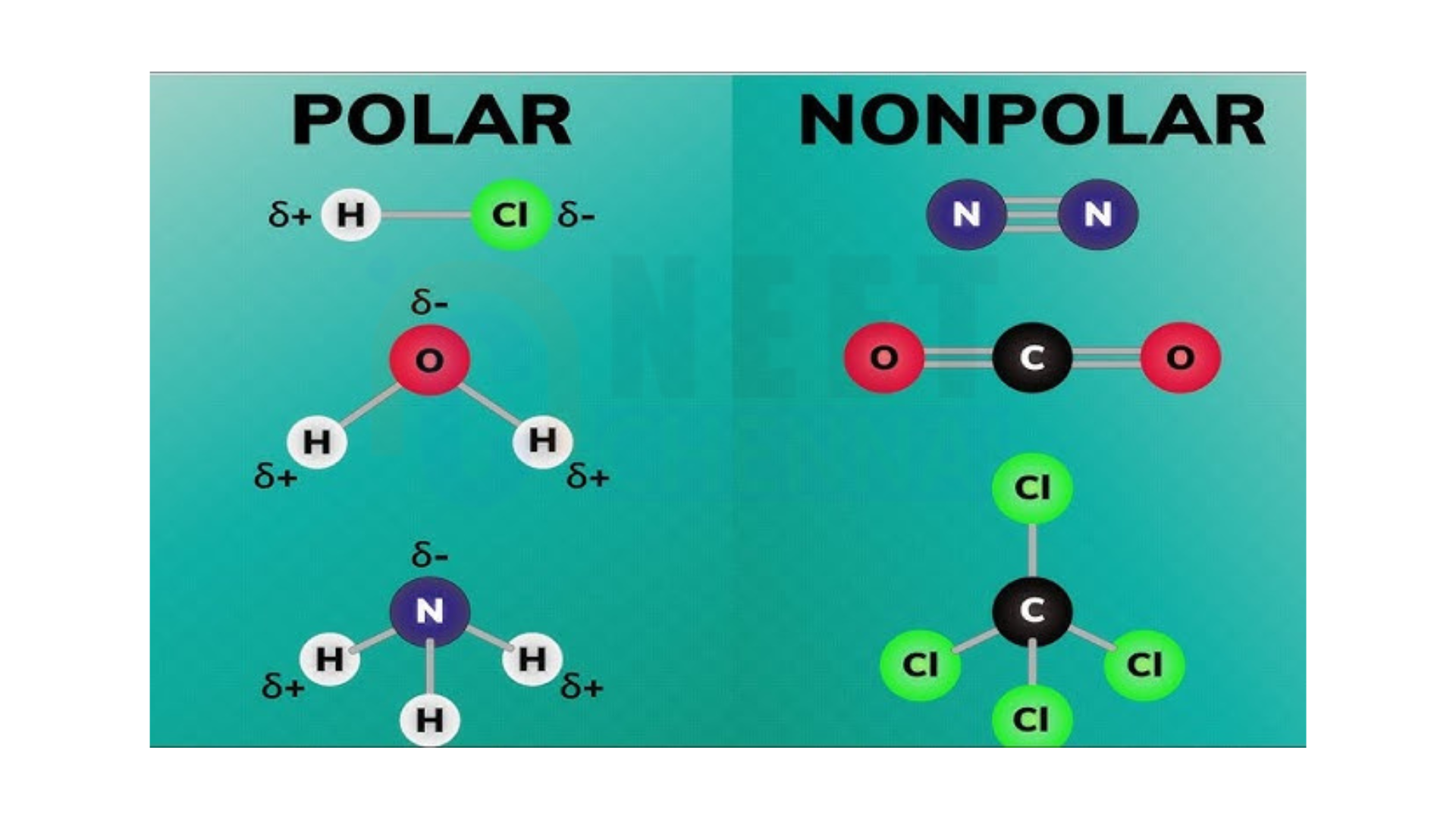
Differentiate between polar and nonpolar dielectric.
Dielectrics are materials that act as electrical insulators and become polarised in an electric field. When exposed to this field, they do not allow current to flow due to the absence of free or loosely bound electrons. Dielectrics can be classified into two categories: polar and nonpolar.
Contents
Key Differences
Feature |
Polar Dielectrics |
Nonpolar Dielectrics |
| Dipole Moment | Possess an inherent dipole moment | Lack dipole moments |
| Charge Separation | Charges are separated when in an electric field | Charges remain unseparated in an electric field |
| Polarity | Display polarity | Do not display polarity |
| Electrical Conductivity | Polar compounds that do not conduct electricity | Nonpolar compounds that also do not conduct electricity |
| Shape | Have asymmetric shapes | Have symmetric shapes |
| Examples | Water and ammonia | Benzene and methane |
Conclusion
Dielectrics can be categorised as either polar or nonpolar based on their molecular characteristics. The primary distinction is in their shape: polar dielectrics have an asymmetric structure, while nonpolar dielectrics are symmetric. Both types serve as insulators but react differently under an electric field.