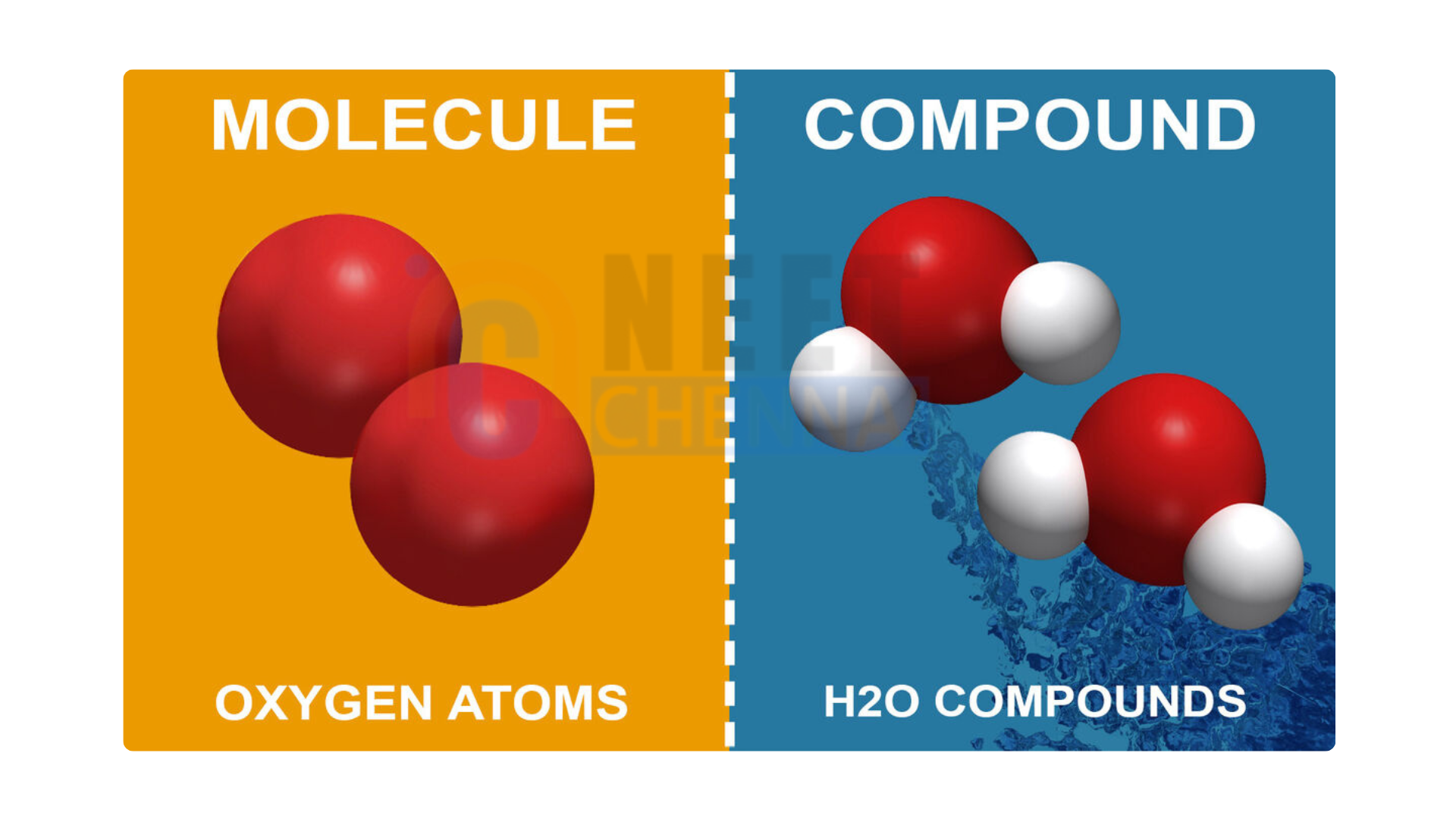
Introduction to Molecules and Compounds
In chemistry, molecules and compounds are fundamental concepts that represent the building blocks of matter. Understanding their differences is essential for grasping key chemical principles and the interactions between substances. This introduction will define molecules and compounds and set the foundation for a deeper exploration of their characteristics and distinctions.
What is a Molecule?
A molecule is the smallest unit of a substance that retains its chemical identity. It consists of two or more atoms that are chemically bonded together, which can be either the same or different elements. For instance, the molecular formula for water is H₂O, which signifies the presence of two hydrogen atoms and one oxygen atom. Molecules can exist independently and possess specific physical and chemical properties.
What is a Compound?
A compound is a type of molecule that consists of two or more different elements chemically bonded in a specific ratio. Compounds are created through chemical reactions, where atoms of various elements combine to form a new substance. Each compound has a unique chemical formula that indicates the ratio of its elements.For instance, sodium chloride (NaCl) is a compound made from one sodium atom and one chlorine atom. Compounds display distinct properties that are different from the individual elements that compose them.
Characteristics of Molecules and Compounds
Characteristics of Molecules
- Composition: Molecules consist of two or more atoms that may be of the same or different elements.
- Size: Molecules can vary significantly in size, ranging from small structures with just a few atoms to large, complex molecules containing many atoms.
- Stability: Generally, molecules are stable and can exist independently, possessing specific chemical properties that allow them to undergo reactions and form new molecules.
- Physical Properties: Molecules exhibit unique physical properties, such as melting point, boiling point, density, and solubility, determined by the arrangement and types of atoms.
- Molecular Formula: Each molecule has a molecular formula that shows the types and quantities of atoms present. For example, the molecular formula for water is H₂O, indicating two hydrogen atoms and one oxygen atom.
Characteristics of Compounds
- Composition: Compounds are formed from two or more different elements that are chemically bonded, with a fixed elemental ratio in their chemical formula.
- Formation: Compounds result from chemical reactions where atoms of different elements combine to create a substance with distinct properties.
- Chemical Properties: Compounds exhibit unique chemical properties that differ from the elements that make them up, allowing them to react with other substances in specific ways.
- Physical Properties: Compounds have characteristic physical properties, including colour, odour, taste, and texture, which are distinct from their constituent elements.
- Chemical Formula: Each compound has a chemical formula that indicates the elements and their ratios. For example, sodium chloride (NaCl) shows one sodium atom and one chlorine atom.
- Stability: Compounds are generally stable and can exist as distinct entities, with properties different from those of their individual elements.
- Energy Changes: Chemical reactions involving compounds often result in energy changes, such as the release or absorption of heat or light.
Key Differences Between Molecules and Compounds
| Sr. No. |
Feature |
Molecule |
Compound |
|
1 |
Definition | A group of two or more atoms bonded together, which may be the same or different elements. | A specific type of molecule made from different elements that are chemically bonded. |
|
2 |
Composition | Can consist of the same or different elements. | Always made from different elements. |
|
3 |
Bonding | Atoms are held together by covalent bonds. | Atoms are bonded by ionic or covalent bonds. |
|
4 |
Representation | epresented by a molecular or structural formula. | Represented by a chemical formula indicating the elements and their ratios. |
|
5 |
Examples | O₂ (oxygen), N₂ (nitrogen), H₂O (water). | H₂O (water), CO₂ (carbon dioxide), NaCl (sodium chloride). |
|
6 |
Properties | Retains properties of the constituent atoms or elements. | Displays unique properties that differ from the elements that compose it. |
Summary
Molecules and compounds are key concepts in chemistry. A molecule consists of two or more atoms connected by chemical bonds and represents the smallest unit of a substance that retains its chemical properties. In contrast, compounds are specific types of molecules made up of different elements, formed when atoms chemically bond in fixed ratios. The primary distinction between molecules and compounds lies in their composition: molecules can contain the same or different types of atoms (e.g., O₂, N₂), whereas compounds are always composed of different elements (e.g., H₂O, CO₂). Compounds have unique chemical formulas and properties, while molecules can be either individual elements or combinations of different elements.