What is Ionic, Covalent and Metallic Bonds: Introduction
Chemical bonds are forces of attraction between atoms to come together and form molecules and compounds. Atoms have a positive charge in the centre, referred to as the nucleus which is made up of protons and neutrons; surrounded by negative electrons. Chemical bonds develop between atoms when they interact and share or even transfer electrons to acquire a more stable electron configuration.
Three Main Types of Chemical Bonds
There are three main types of chemical bonds, including ionic, covalent, and metallic bonds.
Ionic Bond
Ionic bond between atoms with significantly different electronegativities is created. In an ionic bond, one of the atoms transfers its electrons to another atom, thus giving a positively charged ion called a cation, and the other with a negative charge will be called an anion. These oppositely charged ions are held together by electrostatic forces of attraction.
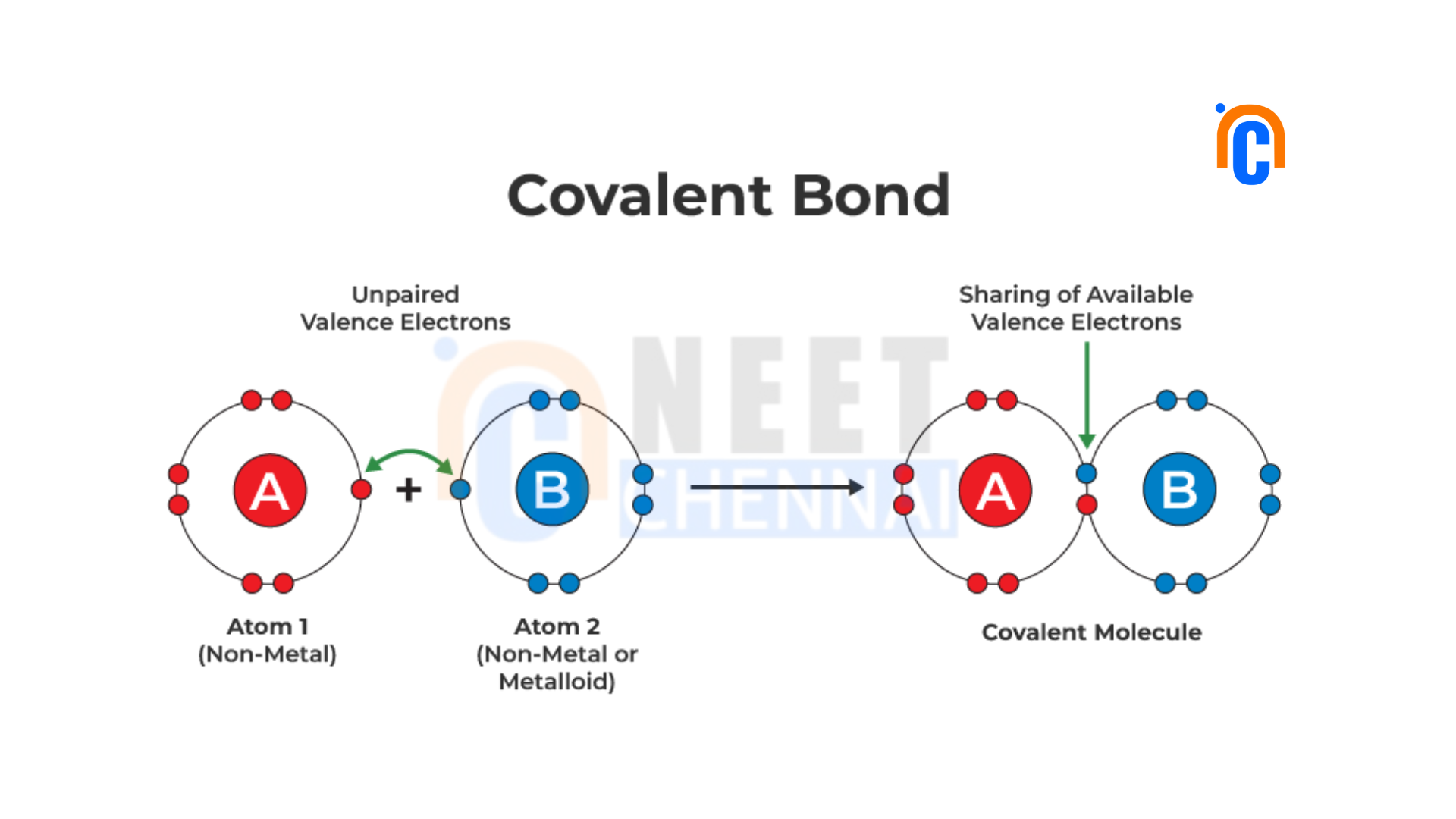
Covalent Bond
A covalent bond is formed between atoms with similar electronegativities. In a covalent bond, atoms share electrons to achieve a more stable electron configuration. The shared electrons form a bond, creating a molecule. Covalent bonds can be further classified as nonpolar covalent bonds and polar covalent bonds, depending on the electronegativity difference between the atoms involved.
Metallic Bond
Metallic bonds are characteristic of metals and are due to the lattice of metal atoms. In a metallic bond, the valence electrons of metal atoms are delocalized and free to roam throughout the lattice. The “sea of electrons” holds the metal atoms together through electrostatic attractions.
Formation of Ionic Covalent and Metallic Bonds
Formation of Ionic Bond
The formation of an ionic bond occurs when one atom in the bond has a much greater electronegativity than the other. Here, the atom which has greater electronegativity will attract the valence electrons belonging to the other atom more strongly, such that those valence electrons are transferred. Transfer leads to the development of ions of opposite charges. The positively charged ion is called a cation and the negatively charged ion is called an anion. These ions then come together through electrostatic forces of attraction to form an ionic compound.
Example: Sodium (Na) donate an electron to chlorine (Cl), forming Na+ and Cl– ions. The strong electrostatic attraction between these oppositely charged ions creates an ionic bond.
Formation of Covalent Bond
The mutual sharing of electrons between atoms forms covalent bonds. The involvement of atoms in covalent bonding results in atoms that possess approximately the same electronegativity. The shared electrons go to the outermost energy levels, which are called valence shells, of both atoms and result in the stable electron configuration for both atoms. This shared pair of electrons forms a covalent bond and holds the atoms together in a molecule.
Example: Water (H2O), Methane (CH4).
Formation of Metallic Bond
Metallic bonds result in metals because of the delocalization of valence electrons. In a metallic lattice, each metal atom donated its valence electrons to a common electron cloud thereby creating a “sea of electrons.” Because of this delocalization, the valence electrons were free to move all over the lattice and accounted for the high thermal and electrical conductivities of metals.
Example: Copper (Cu), Iron (Fe)
Properties of Ionic, Covalent and Metallic Bonds
Properties of Ionic Bonds
Formation of Ions: In electrovalent bonds, one atom of an element loses one or more electrons to another atom forming a cation with positive charge and an anion of negative charge.
High Melting and Boiling Points: Electrovalent compounds have a very high melting point and boiling point owing to the strong electrostatic forces of attraction existing between ions. An electrovalent bond requires much energy to be broken.
Solubility in Polar Solvents: Electrovalent compounds are highly soluble in polar solvents like water. The ionic compounds can easily break into their constituent ions, which then interact with the solvent molecules.
Conductivity Electrovalent compounds are excellent conductors of electricity either in the molten state or when they are dissolved in water. The ions are responsible for the electric current flowing.
Crystal Lattice Structure: Electrovalent compounds typically exhibit a three-dimensional crystal lattice structure. The alternate arrangement of positive and negative ions provides the maximum attractive forces between ions.
Properties of Covalent Bonds
Electron Sharing: In the covalent bond, atoms share electrons. Then an electron cloud in the form of a shared circle appears.
Variable Bond Strength: Covalent bonds may be weak or strong, depending on such factors as bond length and bond energy. Shorter and stronger bonds require more energy to break.
Lower Melting and Boiling Points: In general, covalent compounds exhibit lower melting and boiling points than electrovalent compounds. The intermolecular forces acting between molecules are relatively weaker than the strong electrostatic forces existing within ionic compounds.
Lower Melting and Boiling Points: Covalent compounds are soluble in most nonpolar solvents such as hexane or benzene, as they do not significantly interact with covalent bonds.
Poor Conductivity: Covalent compounds are poor conductors in any state because they have no freely moving charged particles. There are exceptions to this where some covalent compounds would dissociate into ions in a solution and some covalent compounds have mobile charge carriers for example graphite.
Properties of Metallic Bonds
Delocalized Electrons: In metallic bonds, the valence electrons get delocalized, hence free to move within the metallic lattice.
High Thermal and Electrical Conductivity: Delocalization of electrons within metallic bonds enables efficient heat and electrical energy transfer.
Malleability and Ductility: The metallic bonds make metals more malleable and ductile because the free electrons are capable of moving and flowing under stress.
Lustre: Metallic bonds give metals their characteristic shine, or lustre, due to the reflection of light by the free electrons. Variable strength: The strength of metallic bonds is variable, depending on factors such as the size and arrangement of the metal atoms.
Difference Between Ionic Covalent and Metallic Bonds
| Sr. No. |
Characteristic |
Ionic Bond |
Covalent Bond |
Metallic Bond |
| 1 | Formation | Complete transfer of electron from one atom to another | Sharing of electrons between atom | Delocalization of valence electron |
| 2 | Electron Sharing | Minimal or absent | Significant | Extensive |
| 3 | Bond Type | Results in the formation of oppositely charged ions (cation and anion) | Forms a shared electron cloud between bonded atom | Forms a lattice of positive ions in a sea of delocalized electron |
| 4 | Electron Configuration | Achieves a noble gas electron configuration by transferring electron | Achieves a stable electron configuration by sharing electron | Achieves a partially filled or half-filled electron configuration in the valence band |
| 5 | Strength | Generally stronger than covalent bond | Can vary in strength | Can vary in strength |
| 6 | Examples | Sodium chloride (NaCl), Calcium oxide (CaO) | Water (H2O), Methane (CH4) | Copper (Cu), Iron (Fe) |
| 7 | Applications | Medicine, Construction Materials, Fertilizer | Organic Chemistry, Electronics and Semiconductors, Polymers and Plastic | Electrical conductors, Structural materials, and Jewelry |
Summary
In ionic bonds, the electrons are transferred from one atom to another completely. This results in oppositely charged ions that are held together by strong electrostatic forces. Ionic bonds generally occur between atoms with a large difference in electronegativity. However, covalent bonds result from shared electrons between atoms. The sharing of these electrons occurs in a way that leads to a shared electron cloud, which functions to bind the atoms together. Covalent bonds typically form between two atoms with similar electronegativity values. Metallic bonding is characteristic of metals and arises due to delocalisation of valence electrons over a metallic lattice.