An Introduction to Hypotonic and Hypertonic Solutions
Solutions affect the living organisms’ behavior and functioning. Hypotonic and hypertonic solutions are two kinds of solutions that greatly influence cells and living organisms. The solutions have a concentration of solutes and act differently due to their osmotic behavior, which varies greatly among the effects caused to cells and the physiological processes involved. Therefore, knowledge regarding the differences between hypotonic and hypertonic solutions is important to understand cellular responses, medical applications, and environmental adaptations. In this paper, I’ll explain hypotonic and hypertonic solutions based on their peculiar properties, effects on cells, physiological significance, and application in diverse fields of interest.
Characteristics of Hypotonic and Hypertonic Solution
Definition and Composition
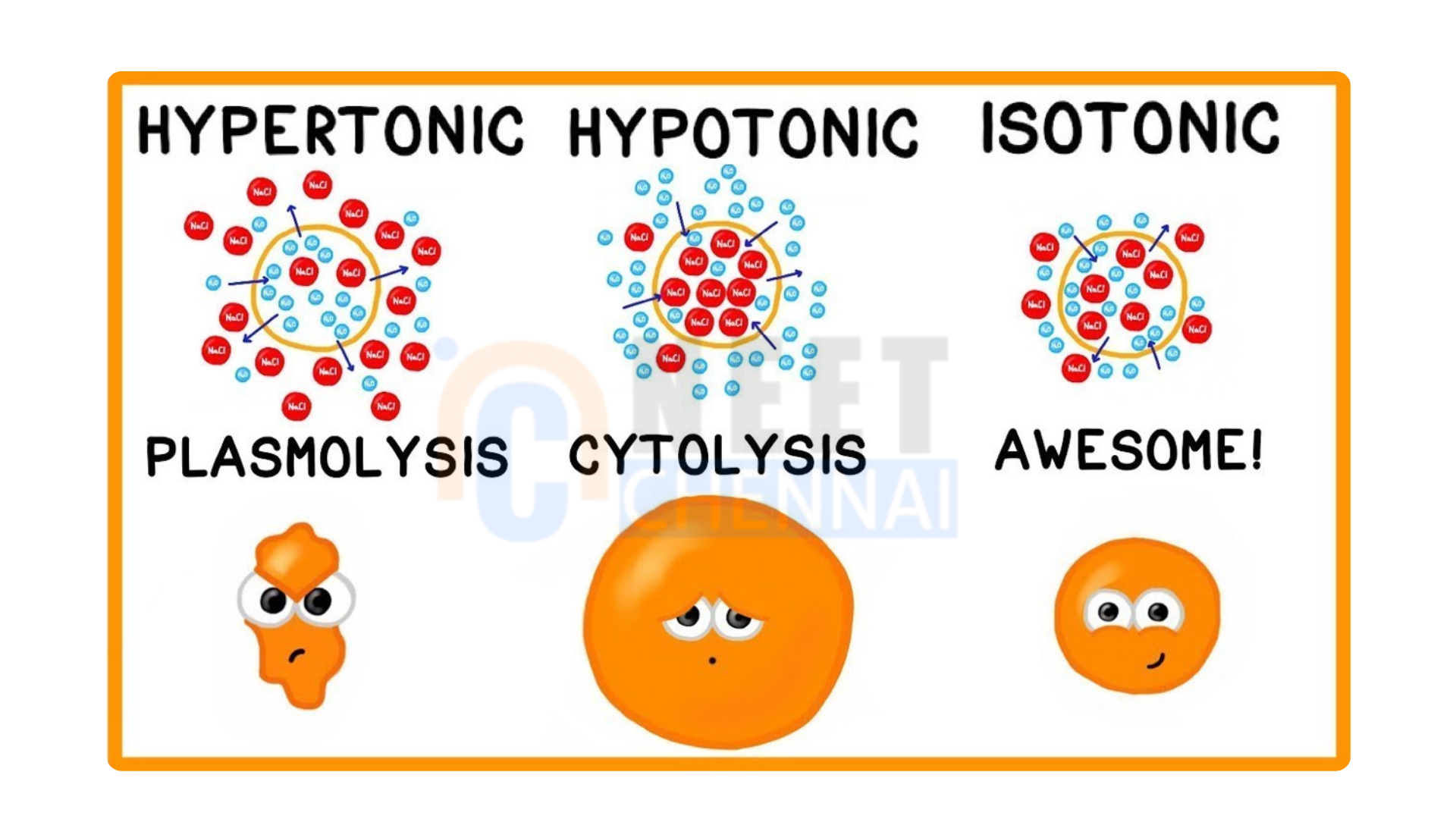
A hypotonic solution is a solution that contains fewer solute concentrations than another solution or the cytoplasm of a cell. This causes a gradient that allows water molecules to enter the hypotonic solution. The nature of the hypotonic solution varies, but it typically contains fewer solute particles than its surroundings.
A hypertonic solution is one that has a higher concentration of solutes than another solution or the cytoplasm of a cell.
Osmosis and Cellular Environment
Since osmosis involves how and when the water molecule moves across a semi-permeable membrane, hypotonic and hypertonic solutions are central to the understanding of the behavior of such a solution.
In the hypotonic solution, there happens to be a larger number of water molecules outside the cell or the solution in comparison with the cytoplasm of the cell. It will result in the movement of water molecules into the cell or hypotonic solution in a way of equalizing the concentrations of the solute on both sides of the membrane. Where one solution tends to be hypotonic, there is always a higher number of water molecules out that the cell than in it. Consequently, water moves into the cell, swelling the cell and can even burst for extreme cases. On the other hand, in a hypertonic solution, there are more water molecules inside the cell. If this were to happen, then water would move out of the cell due to osmosis, causing the cell to shrink or become created.
Effects on Cells and Organisms
Differences in hypotonic and hypertonic solutions have the most fundamental effects on cells and organisms.
Effects of water entry into cells due to hypotonic solutions The cell will swell up because water enters the cell, and ultimately it may break because of bursting known as cell lysis. Inside the cell, the pressure increases due to the entering osmosis resulting in increased stress on the cell membrane.
In hypertonic solutions, the effect is the opposite. The result in animal cells is that water leaves from the cell to enter the hypertonic solution. The cells shrink and may undergo cellular dehydration. Plant cells experience plasmolysis – the cytoplasm shrinks away from the cell wall. This can cause wilting, limiting the plant’s ability to function properly, such as its photosynthetic activities.
Physiological and Medical Implications
Physiologically and medicinally, the hypotonic and hypertonic solutions vary.
APPLICATIONS IN MEDICAL PROCEDURES Such solutions are used in some surgical procedures to irrigate tissues and help hydration of cells. Hypotonic solutions are also used in diagnostic tests like the red blood cell lysis test to test the integrity and functioning of red blood cells. In clinical practice, hypotonic solution 0.45% saline is used to rehydrate patients and replace fluid loss.
On the other hand, hypertonic solutions like 3% saline are used to diminish cerebral enema by forcing out excess water from the cells of the brain. Hypertonic saline is also used for cell shrinking in cancerous tissues and eventually leads to the elimination of those tissues.
Environmental Adaptations
Organisms have evolved various survival strategies under hypotonic and hypertonic conditions.
Organisms living in hypotonic environments have developed several adaptations for maintaining osmotic balance. Such organisms in freshwater, including the freshwater fish, suffer an entry of water into their bodies because the concentration of water is hypotonic to the internal environment of the organism. Freshwater fish counteract such an entry by specialized cells and organs, namely ion pumps and active transport mechanisms that actively regulate the concentration of ions and excrete excess water for achieving osmotic equilibrium.
On the other hand, marine organisms experience a hypertonic environment and suffer from the loss of water. Specialized structures like gills and kidneys have been acquired to actively transport ions and conserve water to balance the osmotic pressure.
Difference Between Hypotonic and Hypertonic Solutions
|
Properties |
Hypotonic Solution |
Hypertonic Solution |
| Solute Concentration | Lower than the surrounding medium or cell cytoplasm | Higher than the surrounding medium or cell cytoplasm |
| Osmotic Movement | Water moves into the cell | Water moves out of the cell |
| Cell Effect (Animal) | Cell expands, may lead to cell bursting (lysis) | Cell shrinks, may lead to cell dehydration |
| Cell Effect (Plant) | Cell becomes turgid (rigid) due to the rigid cell wall | Cell undergoes plasmolysis, cytoplasm shrinks away from the cell wall |
| Physiological Use | Used for rehydration in medical settings | Used to draw excess water from brain cells (cerebral edema) or induce cell shrinkage in cancerous tissues |
| Environmental Adaptation (Freshwater) | Specialised cells and organs to excrete excess water and maintain osmotic balance | Adaptations to actively transport ions and conserve water to prevent water loss |
| Environmental Adaptation (Marine) | Adaptations to actively transport ions and conserve water to prevent water loss | Specialised gills and kidneys to actively transport ions and conserve water |
Conclusion
The greatest difference between these solutions happens in concentration and behavior that takes places towards the solutes once these are exposed to cells and organisms. The hypotonic solution causes cells to expand, while hypertonic solutions cause shrinkage of them. Knowing the effect is very essential to be used in different fields, from biology and medicine to environmental adaptation. Knowing the difference between hypotonic and hypertonic solutions will help researchers, medical personnel, and environmental scientists make intelligent decisions to maintain homeostasis and enhance the well-being of organisms.