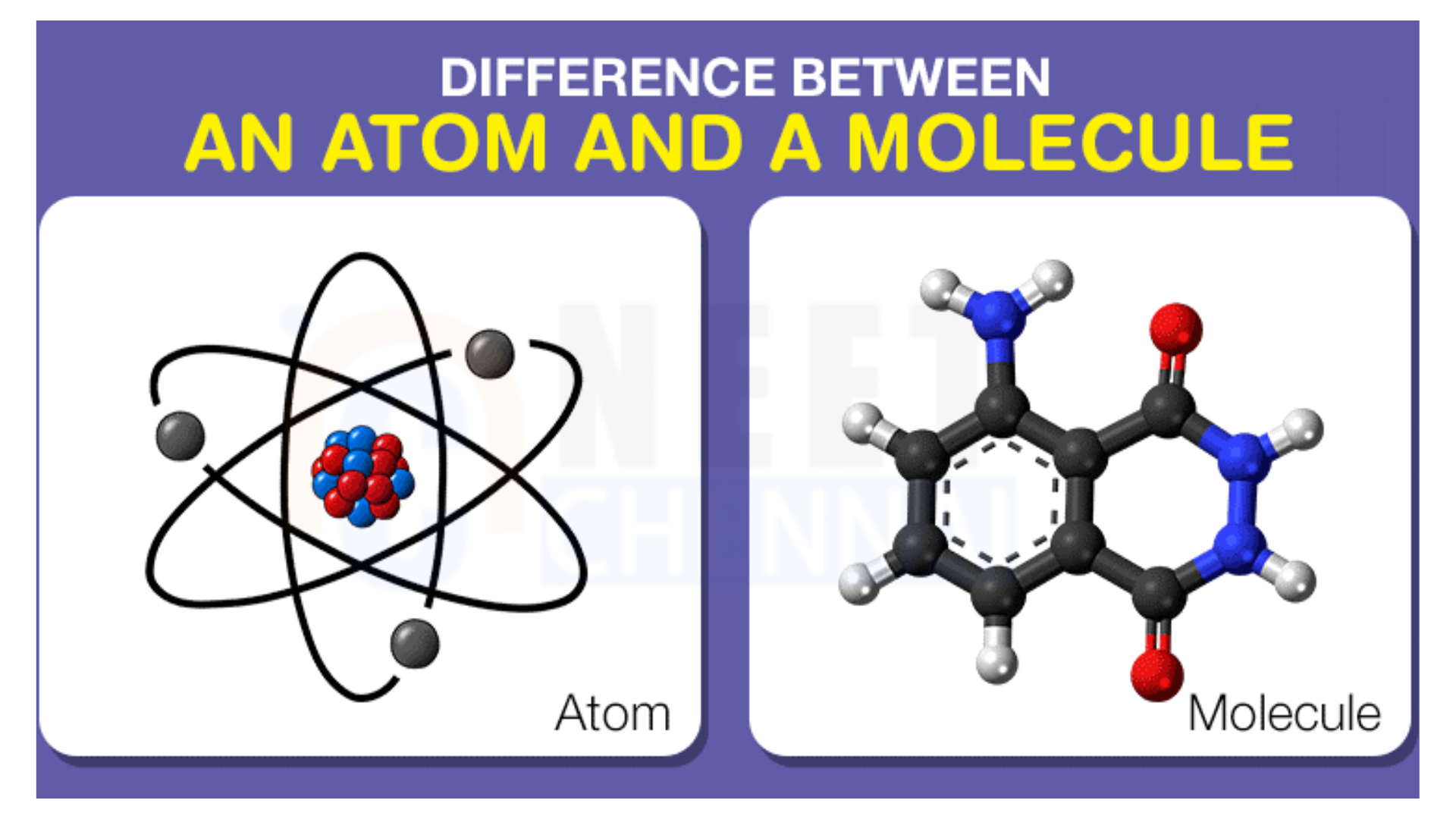
An Introduction to Atoms and Molecules
Atoms and molecules are a constituent building block of matter. What makes them basically different from each other? To know the basic difference between atoms and molecules, we must enter the realm of atomic and molecular structure. Atoms, an indivisible unit of elements, whereas molecules consist of several atoms bonded together. Here in this article, we discuss their contrasting characteristics, composition, and behaviors of this interesting scientific world.
Characteristics of Atoms and Molecules
Atomic Structure
Atoms are therefore the smallest units of matter that retain the characteristics of an element. They consist of the positively charged nucleus of protons and neutrons, along with the cloud of negatively charged electrons surrounding them. The central part is the nucleus, which holds the bulk of an atom’s mass; electrons are found in the electron shells or energy levels around this nucleus. Since the number of protons determines which element the atom is, atoms possess an atomic number. As the electrons and protons oppose each other through a negative and positive charge respectively, atoms are electrically neutral.
Molecular Structure
Two or more atoms come together to form a molecule. Chemical bonds in the form of covalent, ionic, and metallic chemistry hold these atoms together. The bond in a covalent is one in which the atoms share their electrons with each other to form the stable molecular structure. Ionic bonds are the ones in which the transfer of electrons between the atoms results in the formation of charged particles called ions. Metallic bonds are typical of metals in which the delocalized electrons create a “sea” holding the atoms together.
Composition and Types
Atomic Composition
Atoms represent the basic building blocks of all elements within a periodic table. Different atoms along with their types are specific to each element’s atomic number. For example, atoms of hydrogen compose hydrogen (H), and the atoms of oxygen comprise oxygen (O), and so on. Elements can be found in either the free state or in the combined state with other elements that form a compound.
Molecular Composition
By definition, a molecule is made of two or more atoms. The atoms making up a molecule can be the same or they can be different. For instance, oxygen gas (O₂) is a molecule formed by two oxygen atoms chemically bonded together. Water, H₂O, is another type of molecule, and it consists of two hydrogen atoms combined with one oxygen atom. Compounds such as carbon dioxide, CO₂, and sodium chloride, NaCl, are made up of different types of atoms chemically bonded together.
Behavior and Interactions
Atomic Behaviour
Atoms and molecules differ in structure and therefore differ also in behavior. Atoms are relatively stable and do not readily react to one another. However, atoms can react through chemical reactions, at which time atoms can lose, gain, or share electrons in order to achieve a more stable electron configuration.
Molecular Behaviour
Because the molecules consist of bonded atoms, they are also capable of undergoing chemical reactions. The type of reaction depends on the kind of atoms taking part and their bonding patterns. It is possible for them to react to form a new compound, break up into their constituent atoms, or reform in a different pattern of atomic arrangement.
Difference Between Atoms and Molecules
This table provides a concise overview of the difference between atoms and molecules.
|
Property |
Atoms |
Molecules |
| Composition | Single, indivisible units of an element | Two or more atoms chemically bonded together |
| Structure | Consist of a nucleus (protons and neutrons) surrounded by electrons | Formed through chemical bonds between atoms |
| Charge | Neutral (equal number of protons and electrons) | Neutral (overall charge depends on the atoms involved) |
| Behavior | Relatively stable, undergo chemical reactions to achieve stability | Exhibit a wide range of chemical reactions and interactions |
| Examples | Hydrogen (H), Oxygen (O) | Water (H₂O), Carbon Dioxide (CO₂) |
| Size | Atoms are smaller than molecules | Molecules are larger than atoms |
Conclusion
The underlying difference between atoms and molecules lies in their respective formation and actions. Atoms that basically comprise elements are made up of two or more atoms bound together chemically to form molecules. The periodic table is formed of atoms, each having a unique atomic number, while molecules constitute compounds, hence showing an extensive range of chemical reactions and interactions. Understanding the concept of atoms and molecules helps one make sense of the complexity that is chemistry, the reasons why substances behave the way they do, and what happens at the atomic and molecular level.