Introduction to Apoenzyme and Holoenzyme Difference
To grasp the distinction between an apoenzyme and a holoenzyme, it’s essential to first understand enzymes, their roles, and the concept of cofactors. Let’s explore these subjects in detail first.
What are Enzymes?
Enzymes, which are proteins, act as biological catalysts that accelerate chemical reactions. The substances that bind to enzymes are called substrates, and the enzyme transforms these substrates into different molecules referred to as products. Most cellular metabolic processes require enzyme catalysis to occur quickly enough to support life. Although a few enzymes consist of RNA molecules, the majority are protein-based.
Why are Enzymes Needed?
RNA molecules are responsible for translating genetic information from DNA into proteins. Every cell contains numerous enzymes that the body utilizes for different functions. Enzymes facilitate the chemical reactions essential for sustaining an organism’s health. For example, they play a key role in metabolism, which converts food and liquids into energy.
What are Cofactors?
Many enzymes require a small additional molecule called a cofactor to assist in catalytic activity. A cofactor is a non-protein molecule that helps perform chemical reactions that the body’s standard 20 amino acids cannot accomplish. Cofactors may be small organic compounds, such as coenzymes, or inorganic substances like metal ions.
Functions of Cofactors
Cofactors, both inorganic and organic, act as catalysts in enzymatic reactions, often taking the form of metal ions or coenzymes. Most coenzymes are water-soluble, non-protein organic compounds derived from vitamins through phosphorylation. Coenzymes bind to apoenzyme protein molecules to form active holoenzymes.
What is Apoenzyme and Holoenzyme?
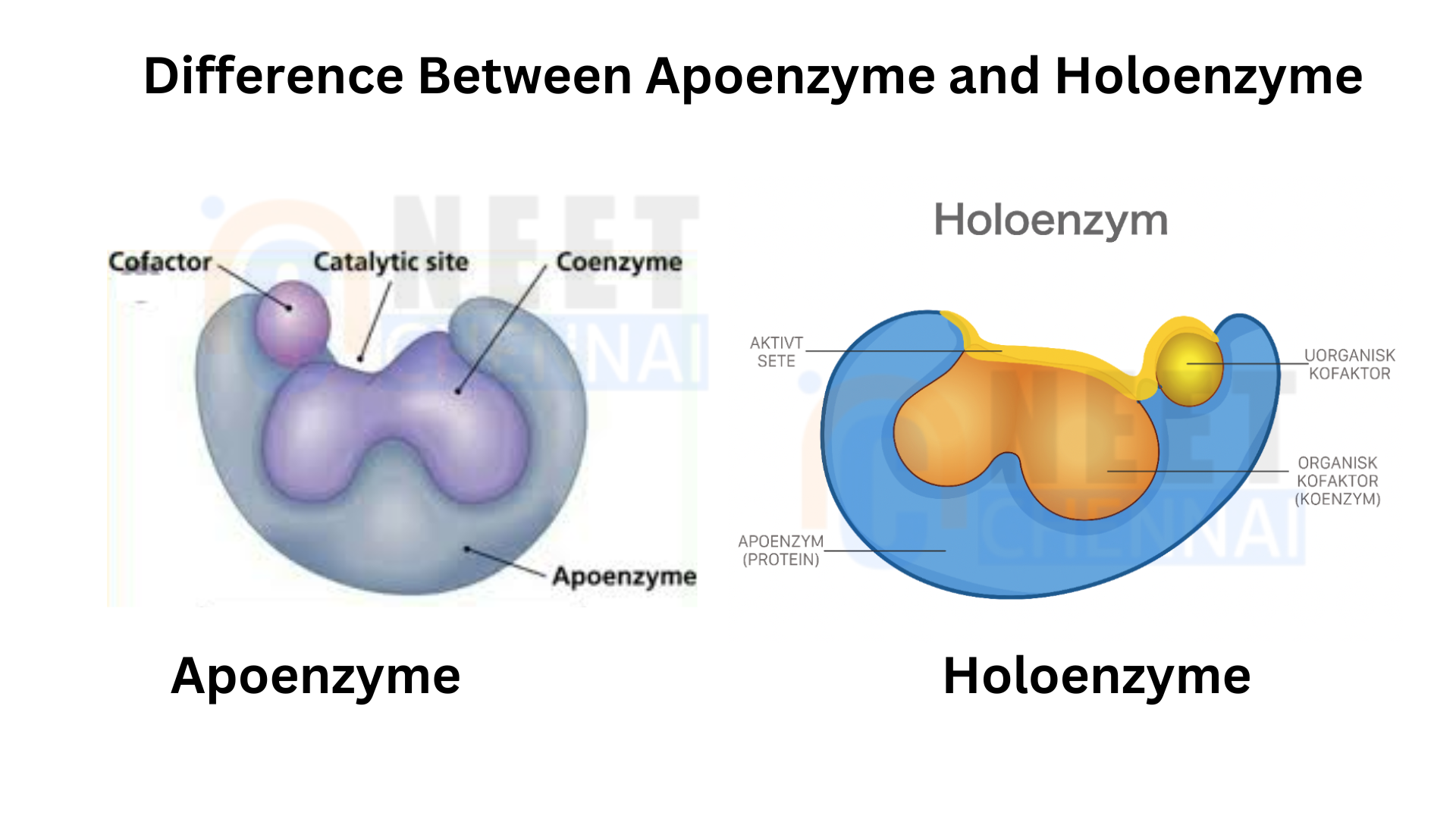
Apoenzyme: An apoenzyme is an enzyme that lacks the necessary cofactor to function properly. Apoenzymes are inactive by themselves but become active once they bind to either an organic or inorganic cofactor. They play a key role in determining an enzyme’s substrate specificity, making them vital for enzymatic activity. Examples of enzymes that contain both apoenzymes and coenzymes include transferases, oxidoreductases, ligases, and isomerases.
Holoenzymes: A holoenzyme is a fully functional, catalytically active enzyme. It forms when an apoenzyme binds to its required cofactors. A holoenzyme contains all the necessary elements required for the enzyme’s function, including enzymes like DNA polymerase III and RNA polymerase.
Apoenzyme(inactive) + Cofactor= Holoenzyme(active).
A holoenzyme is also referred to as a conjugate enzyme. The enzyme’s protein part, called the apoenzyme, remains inactive in the absence of its cofactors.
Therefore, the term for the catalytically active combination of apoenzyme and cofactor is the holoenzyme or conjugate enzyme.
Some Functions of both Apoenzyme and Holoenzyme Include-
- A holoenzyme relies on apoenzymes to function properly. One of the most important holoenzymes is DNA polymerase, which is composed of an apoenzyme and a cofactor.
- DNA polymerase catalyzes the polymerization of deoxyribonucleotides to form DNA. By reading the existing DNA strand, it generates new DNA strands, facilitating DNA replication. It produces a new strand that complements the template strand and is identical to its paired strand. Magnesium ions act as a substitute cofactor for DNA polymerase.
- RNA polymerase is another holoenzyme composed of an apoenzyme and cofactor. It is responsible for transcription, synthesizing RNA chains using DNA templates.
Difference Between Apoenzyme and Holoenzyme
|
S.No |
Category | Apoenzyme |
Holoenzyme |
| 1 | Definition | The catalytically inactive protein in an enzyme. | The combination of an apoenzyme and a cofactor that is catalytically active. |
| 2 | Activity | Initially inactive, it becomes active only upon binding with a cofactor. | Functioning effectively and fully capable of promoting a biological reaction. |
| 3 | Composition | Only the protein constituent is included in apoenzymes. | It include cofactors like metal ions or other chemical complexe called coenzyme together with protein. |
| 4 | Cofactors | There are no cofactors present. | Cofactors like metal ions or coenzymes are present. |
| 5 | Examples | The protein component of the holoenzyme, such as carbonic anhydrase in the absence of the zinc ion (cofactor). | Carbonic anhydrase, DNA and RNA polymerases, etc. |
Summary
Enzymes exist in two forms: apoenzyme and holoenzyme. The main difference between them is that holoenzyme is the catalytically active form, made up of the apoenzyme and its cofactor, while apoenzyme is the inactive protein portion of the enzyme. A cofactor, which can be a metal ion or a small organic molecule, plays a key role by interacting with the apoenzyme’s structure to help facilitate the enzyme’s function.