What are Adhesive and Cohesive Forces?
Cohesive and adhesive forces are basic concepts in physics and chemistry that govern how substances act. Cohesive forces are attraction forces between the molecules of the same substance, while adhesion is an attraction force between two different substances’ molecules. A common daily example of this is observed in the curved upper surface of the tea which has formed at the rim of a glass because of the cohesive forces prevailing in the liquid as well as the adhesive forces between the liquid tea and the inner glass surface. Those forces will also explain such phenomena as surface tension and material adhesion.
Adhesive Force:
Adhesive Force:
The forces of adhesion are the attractive forces between molecules of different substances. That is, the molecules of one substance stick to or adhere to the molecules of another substance. They cause phenomena such as capillary action where the liquid ascends or descends against gravity along a narrow tube.
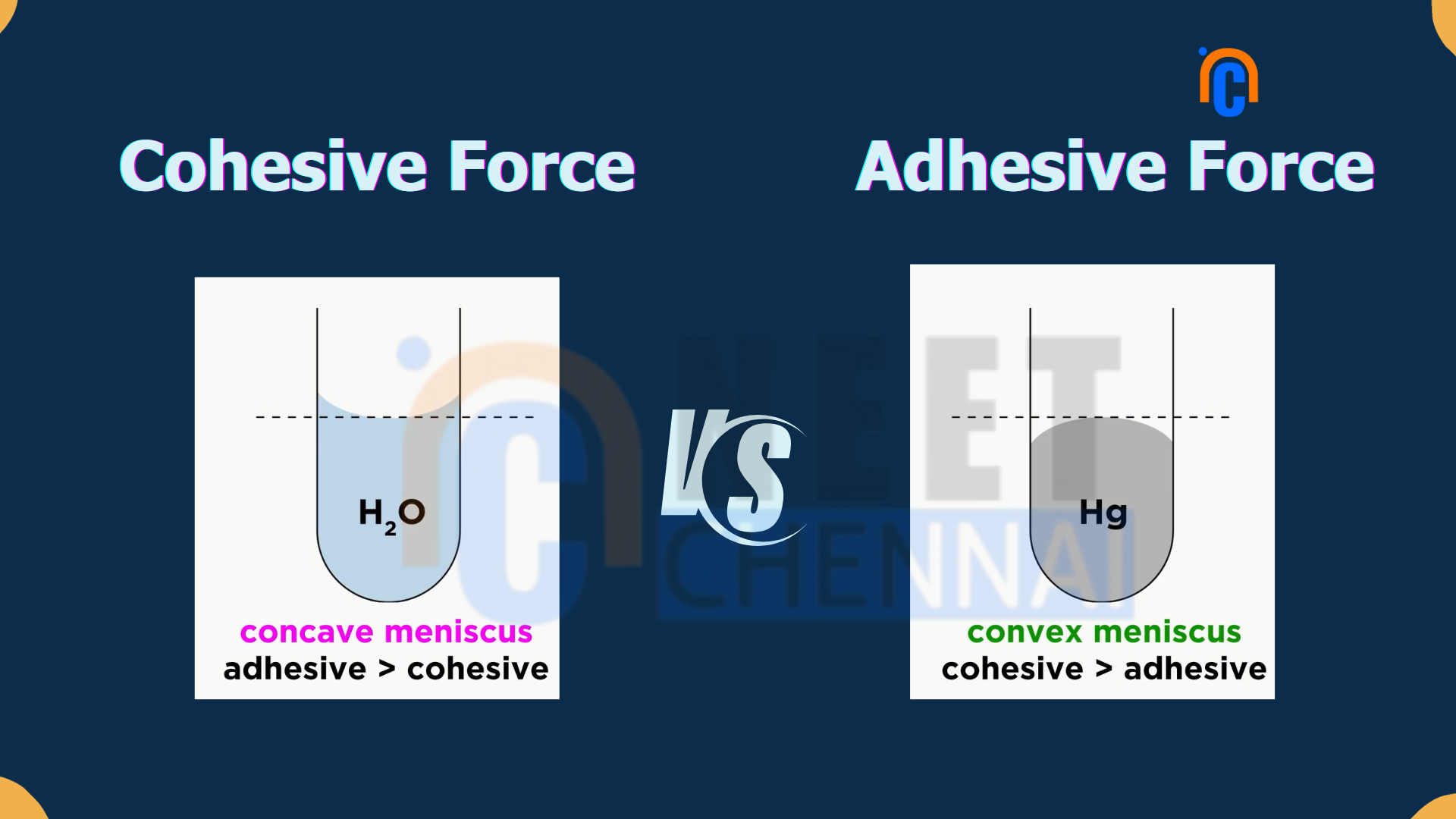
For example, adhesions account for the stickiness of a column of water on the inside walls of a glass tube. It is because of adhesions between water molecules and the surface of the glass that a column of water sticks through the inside walls of the tube against gravity, producing the meniscus, or curved surface, you may have seen in graduated cylinders.
Cohesive Force:
Cohesive forces are attractive forces between molecules of the same substance. These forces hold the molecules together and give rise to the cohesive properties of the material. The state in which matter takes with respect to whether it is solid, liquid, or gas will depend on the strength of the cohesive forces holding the molecules together.
A simple example of cohesive forces can be derived from liquid water. In such cases, the water molecules interact through hydrogen bonds and thereby develop a strong force of cohesiveness. This is why water forms droplets and also displays surface tension. The high value of surface tension in water allows some insects to walk on its surface without sinking, like the water strider.
Comparison between Adhesive and Cohesive Forces:
Understanding the distinction between adhesive and cohesive force is important in various scientific fields, as they have significant implication for phenomena such as adhesion, surface tension, and material behaviour.
The table describes the comparison between adhesive and cohesive forces:
|
Adhesive Force |
Cohesive Force |
| Adhesive force is an attractive force between molecule of different substances. | Cohesive force is an attractive force between molecule of the same substance. |
| Adhesive forces occur between molecule of different substances, allowing them to stick or adhere to each other. | Cohesive forces occur between molecule of the same substance, this force binds the same molecules. |
| Adhesive force are responsible for adhesion and bonding between different materials. | Cohesive force determine the cohesive properties of a substance, such as its ability to maintain its form and flow. |
| Adhesive force contribute to the shape of the meniscus, which is the curved surface of a liquid in a container. | Cohesive force also influence the meniscus, as they determine the curvature and behavior of the liquid’s surface. |
| Adhesive force can be observed when water adheres to the walls of a glass container, or when glue sticks two surfaces together. | An example of cohesive force is observed in liquid water, where hydrogen bonds between water molecules create a strong cohesive force, allowing water to form droplets and exhibit surface tension. |
Reactions of Adhesive and Cohesive Forces
What are the adhesive and the cohesive force reactions? This plays an important role in most of the processes that take place naturally and artificially. These are realized by the intermolecular forces between the molecules, leading to numerous phenomena – adhesion of the materials, droplet formation, and behavior of liquids and solids. Here, we have a number of examples of adhesive and the cohesive force reactions, and we will continue further about them.
Let’s explore the reactions of adhesive and cohesive forces:
Adhesive Force Reaction:
The adhesive force reactions result from the interlinking between molecules of two substances. Once the molecules interlink to produce strong bonds, then, it leads to adhesive force reactions. In our daily lives, such an adhesive force reaction is very common. For example, whenever we try to stick two surfaces together by using glue, adhesion forces between glue and surfaces help in firm bonding of these two surfaces together. Similarly, adhesive forces make tape stick onto paper or on walls temporarily and helps in temporary bonding. Adhesive forces are once more at work when water wets the sides of glass, thereby leaving a meniscus.
Adhesive forces between materials depend on their strength; the stronger the adhesive forces are, the tighter and more durable the bond between materials will be. The weaker the adhesive forces are, the weaker the force holding materials together is likely to be.
Cohesive Force Reaction:
Cohesive forces are known to keep molecules of a given substance tied up in an invisible bond, giving the substance its cohesive nature. Cohesive force reactions are said to happen whenever the molecules within the substance get into contact with each other and arrange themselves in stable combinations.
One of the most common examples of cohesive force reactions can be found in liquid water. Due to its hydrogen bonds, cohesiveness between the water molecules is strong and tightly connected. Cohesion allows it to drop into droplets, shows surface tension, and remains in the liquid state.
Cohesive force reactions account for the behavior of liquids and solids. For example, it is because of cohesion between the particles that solid material owes strength and integrity. For liquid substances, cohesive forces might explain properties such as viscosity and maintenance of a specific shape.
Conclusion:
Adhesive and cohesive forces are essentially concepts explaining the aspect of how different substances attach and how molecules within a substance hold together. In this regard, adhesive forces refer to those forces that act as glue, sticking materials together, such as the glue or tape we stick things together with. Cohesive forces, on the other hand, are those forces that hold the molecules together within the same substance, giving the material specific properties. Understanding these forces proves precious in areas such as biology and engineering, as they affect such things as how cells adhere to one another or how liquids react. It can help scientists make advances both in the area of materials and in the area of technology by researching the interaction of adhesive and cohesive forces.