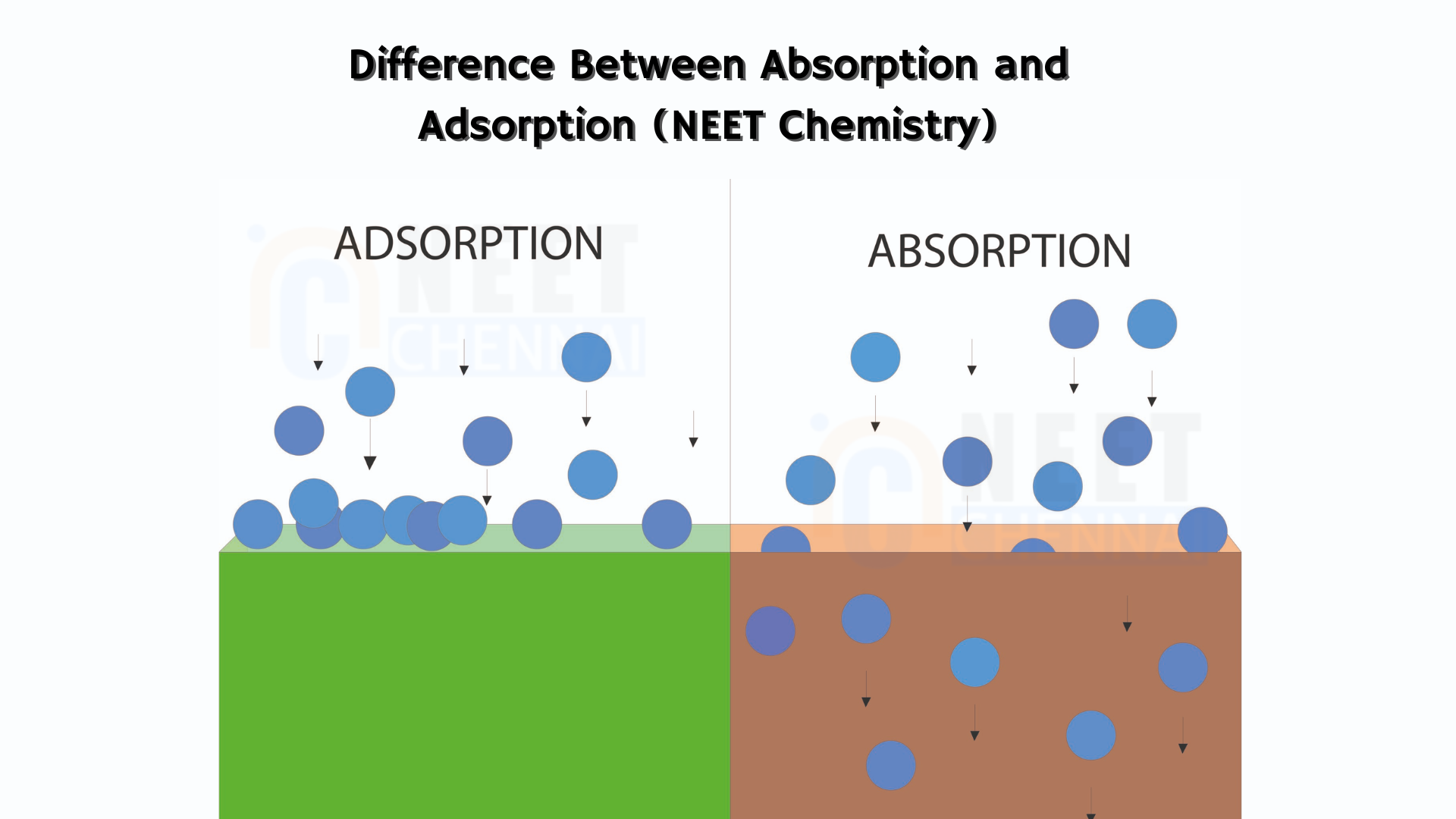
Hint: We understand that absorption and adsorption are distinct processes. In absorption, atoms penetrate into the bulk phase of a liquid or solid, whereas in adsorption, substances attach to the surface of the molecule.
Complete step by step answer:
Let’s explore the key differences between absorption and adsorption.
|
Absorption |
Adsorption |
| Absorption refers to a process where atoms, ions, or molecules are taken up by another substance, typically within a solid or liquid material. | Adsorption is the condition in which gas, liquid or dissolved solids loosely stick to the surface of another substance especially in solid or liquid. |
| It is a bulk phenomenon | It is a surface phenomenon |
| Absorption reaction occurs at a uniform reaction rate | In an adsorption process, the reaction rate gradually rises and eventually reaches equilibrium. |
| Absorption is an endothermic process | Adsorption is an exothermic process |
| There is no change in concentration. | In the Adsorption reaction the Concentration at the bottom of the absorbent is different from the bulk |
| The temperature does not affect the absorption process. | Adsorption occurs at a lower temperature. |
| The Absorption process is applied on cold storage, ice production, and in turbine inlet cooling | The adsorption process is applied in air conditioning, water filtration, and the production of resins. |
Note:
It is important to understand that the absorption process takes place in both living and nonliving organisms to absorb nutrients and maintain internal balance in response to environmental changes. On the other hand, adsorption is employed in separation methods, such as ion-exchange chromatography, to isolate molecules by selectively transferring the liquid phase onto the surface of a solid.