Hint: The Jahn-Teller effect pertains to the distortion of a nonlinear molecular system. It accounts for the decrease in symmetry and energy of the system. This effect arises from the varying degrees of interaction between the ligands and the metal’s d orbitals.
Complete answer:
A nonlinear molecular system experiences distortion, which leads to a reduction in both its symmetry and energy. This phenomenon is called the Jahn-Teller effect.
In octahedral complexes, the axial bond lengths may differ from the equatorial bond lengths, showing the Jahn-Teller effect. The axial bond lengths can be either longer or shorter than the equatorial bond lengths. The Jahn-Teller effect is also observed in tetrahedral complexes. This effect is influenced by the electronic state of the system.
Consider a nonlinear complex. If the electronic configuration in the ground state has degenerate orbitals, the complex will distort to remove this degeneracy and achieve a lower energy state.
In an octahedral field, the metal’s d orbitals split into two energy levels: \(t_{2g} \) and \( \text{e}_g \). The \(t_{2g} \) orbitals are lower in energy and oriented between the axes, avoiding direct interaction with the approaching ligands. The \( \text{e}_g \) orbitals, which are higher in energy, are aligned along the axes, directly facing the incoming ligands.
The interaction between ligands and \( \text{e}_g \) orbitals is greater than that between ligands and \(t_{2g} \) orbitals, high spin \( \text{d}^4 \), low spin \( \text{d}^7 \) and \( \text{d}^9 \)configurations have greater Jahn Teller effect.
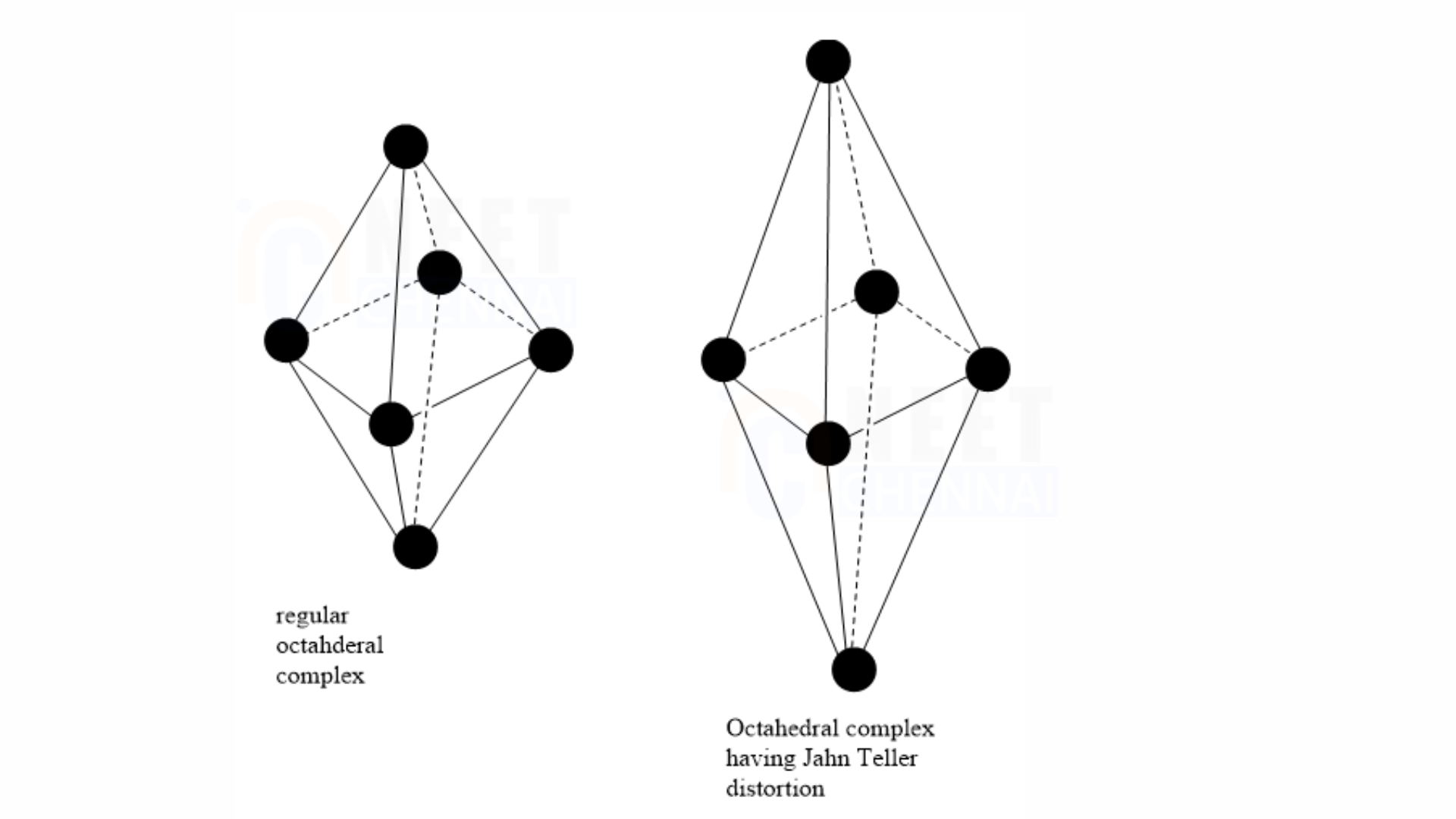
Note: In high-spin \( \text{d}^4 \) octahedral complexes of Cr²⁺ and Cu²⁺, the Jahn-Teller effect occurs as the lone pair of electrons causes the degenerate \( \text{e}_g \) level to split into the \(\ d_{z^2}\) and \( \ d_{x^2 – y^2} \) orbitals.