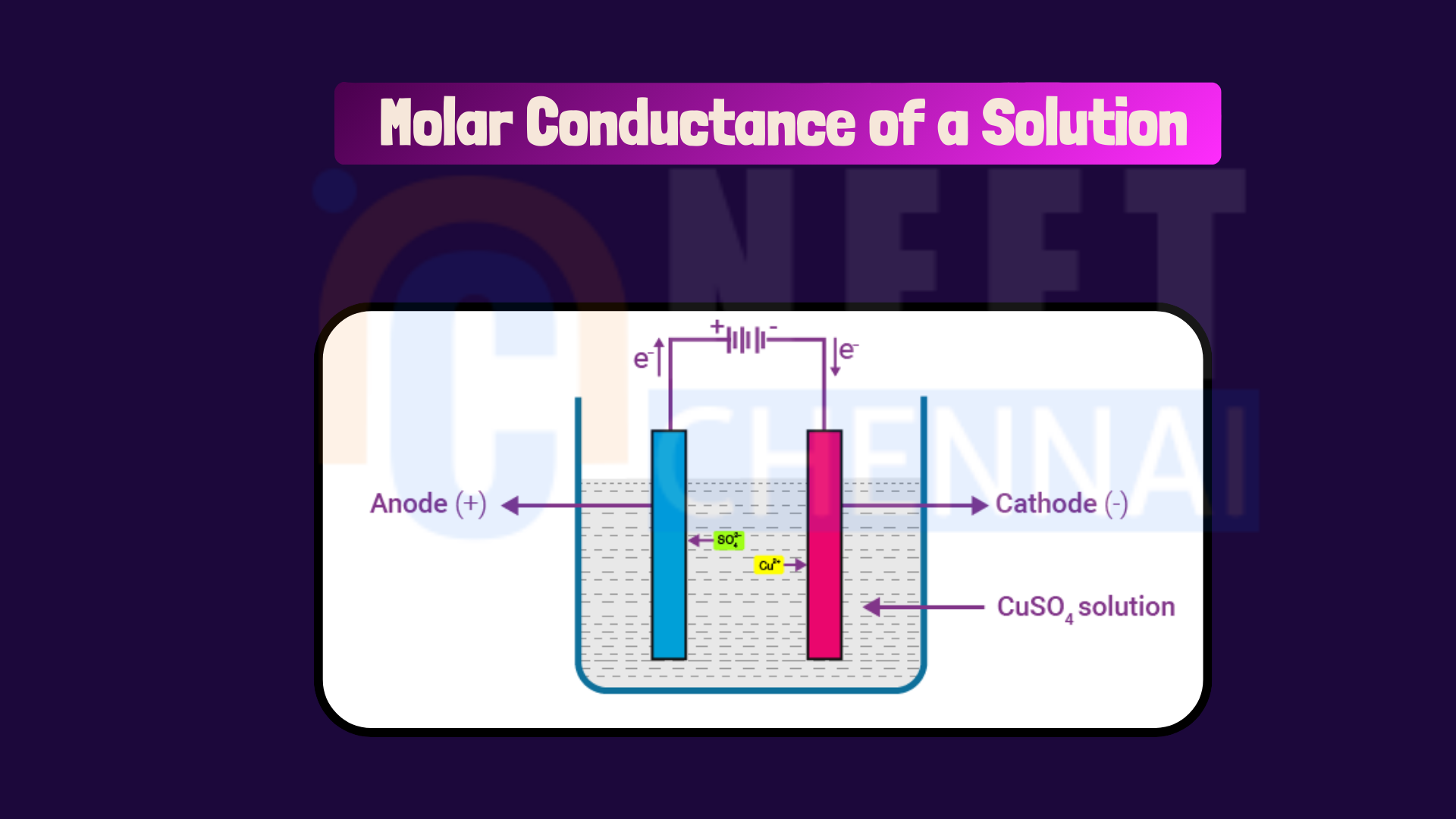
Define molar conductance of a solution. State its unit. How is it related to specific conductance of a solution?
– Hint: The conductance is the property of the conductor (metallic as well as electrolytic) which facilitates the flow of electricity through it. It is equal to the reciprocal of resistance, that is,
Conductance = 1/Resistance =\(\frac{1}{R}\)
There are three kinds of conductance that we take into consideration:
1. Specific Conductance or Conductivity
2. Equivalent Conductance
3. Molar Conductance
Complete step-by-step solution –
The molar conductance of a solution is defined as the conductance of all the ions produced by ionization of 1 g-mole of an electrolyte when present in V ml of solution. It is denoted by \(\wedge _{m}\).
Therefore,
Molar conductance \(\wedge _{m}\) = k * v
where,
V is the volume in mL containing 1 g-mole of the electrolyte. If c is the concentration of the solution in g-mole per litre, then
The unit of molar conductance is ohm−1cm2mol−1
The SI unit of molar conductance
= siemens per metre per (mol per litre)
=S m−1(mol L−1)−1
=S m−1L mol−1
(1 litre = 1000 cm3= 1 dm3= 10−3m3)
∴Units of molar conductance \(\wedge _{m}\)
=Sm−110−3m3mol−1
=10−3S m2mol−1
=(mS) m2mol−1
1 millisiemen (mS) = 10−3S
The general SI unit of molar conductance ( \(\wedge _{m}\))
= S m2mol−1
1 S m2mol−1= 104S cm2 mol−1
or1 S cm2mol−1= 104S m2 mol−1
The following expression is used to mathematically represent the relationship between molar conductance and specific conductance of a solution.
\(\wedge _{m}=\frac{k}{c}\)
where,
= molar conductivity
k = specific conductivity
c = concentration in mole per litre.
Note: Molar conductivity is generally defined as the ability to conduct electricity by all ions produced when one mole of an electrolyte dissolves in solution. It can also be described as the ionic strength of a solution or the salt concentration.