In benzene, the count of sigma and pi bonds is:
a. 6 sigma and 3 pi bonds
b. 12 sigma and 3 pi bonds
c. 9 sigma and 3 pi bonds
d. 6 sigma and 6 pi bonds
Hint: Benzene is a hydrocarbon containing six carbon atoms linked by covalent bonds in a ring structure. Its stability comes from three alternating double bonds within the ring, which make it highly resilient.
Complete step by step answer:
Benzene is an aromatic molecule made of carbon and hydrogen, with the molecular formula C₆H₆. Now, let’s illustrate the structure of benzene.
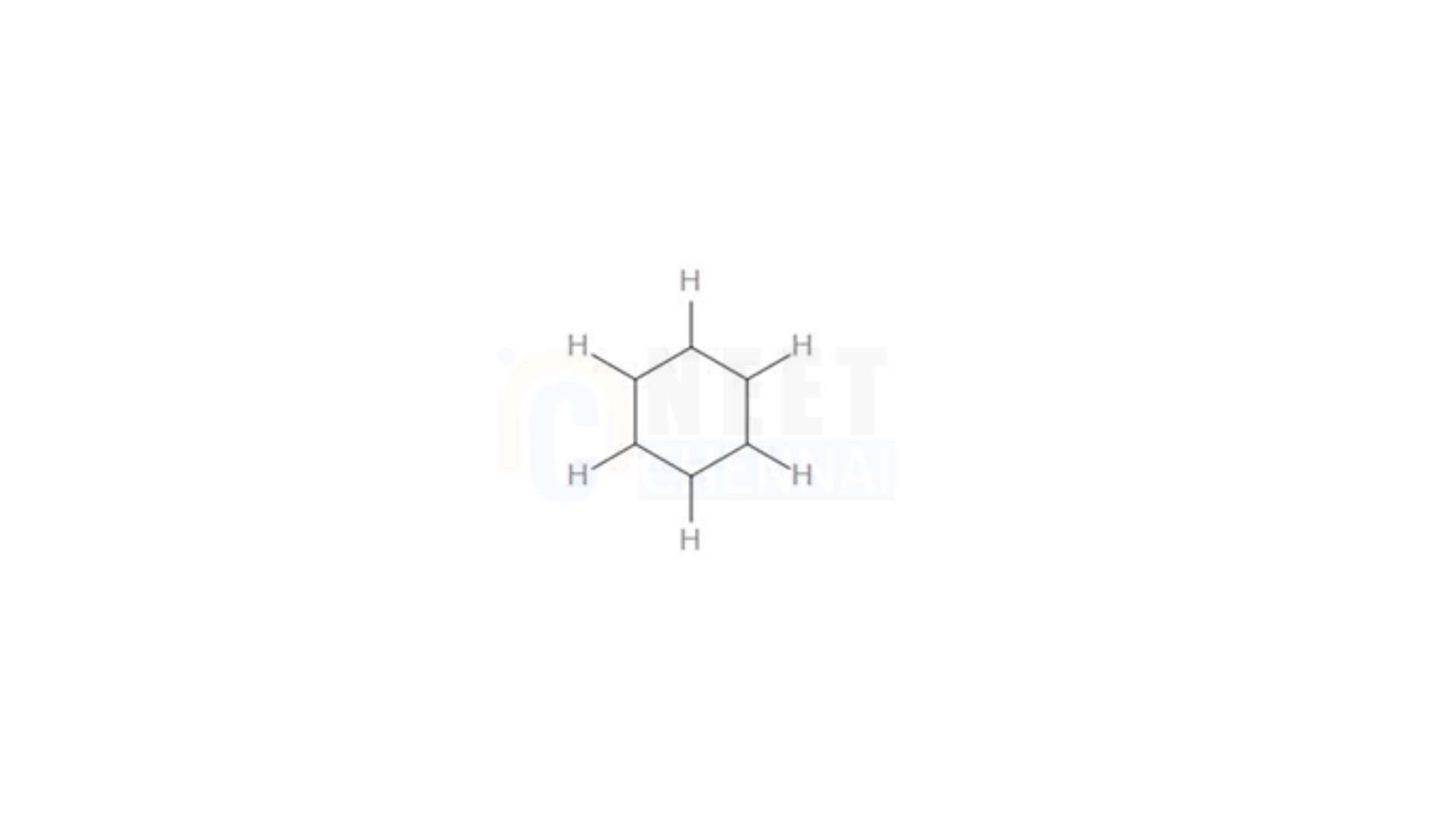
To determine the number of sigma bonds here, we can sketch the skeletal structure, focusing solely on the sigma bonds –
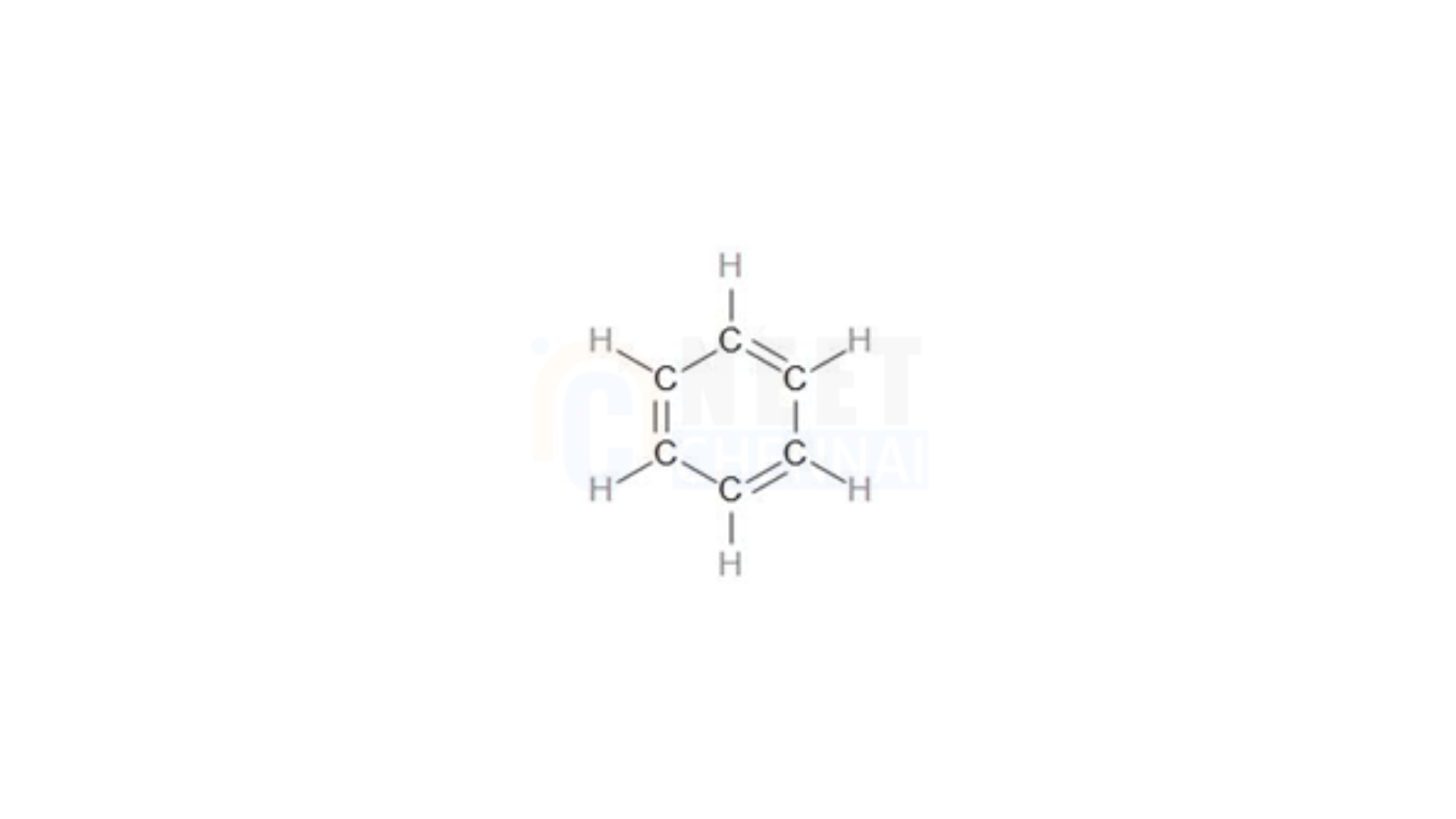
As we observe, benzene contains 6 C-H bonds and 6 C-C bonds. Thus, there are a total of 12 sigma bonds in the structure. Additionally, looking at the benzene structure, we see 3 C=C bonds, which means there are 12 sigma bonds and 3 pi bonds in total. Therefore, benzene consists of 15 covalent bonds.
So, the answer is – option (b) – The number of sigma and pi bonds in benzene are 12 and 3, respectively.
Additional Information: A single bond includes one sigma bond, a double bond has one sigma and one pi bond, and a triple bond consists of one sigma bond and two pi bonds.
Note: Benzene is a colorless, highly flammable liquid with a distinctive sweet odor, which results from the compound’s aromatic nature. This aromaticity in benzene is due to the uninterrupted cycle of pi bonds among its carbon atoms, giving it high stability. Naturally occurring in crude oil, benzene is also a fundamental petrochemical.