How do you convert benzoic acid to benzaldehyde?
Hint: The explanation is that the carboxylic group does not convert directly into the aldehyde group; instead, it first forms an acyl chloride, which is then reduced using Lindlar’s catalyst.
Complete step by step solution:
In earlier sections of organic chemistry, we discussed a reduction reaction known as Rosenmund’s reduction.
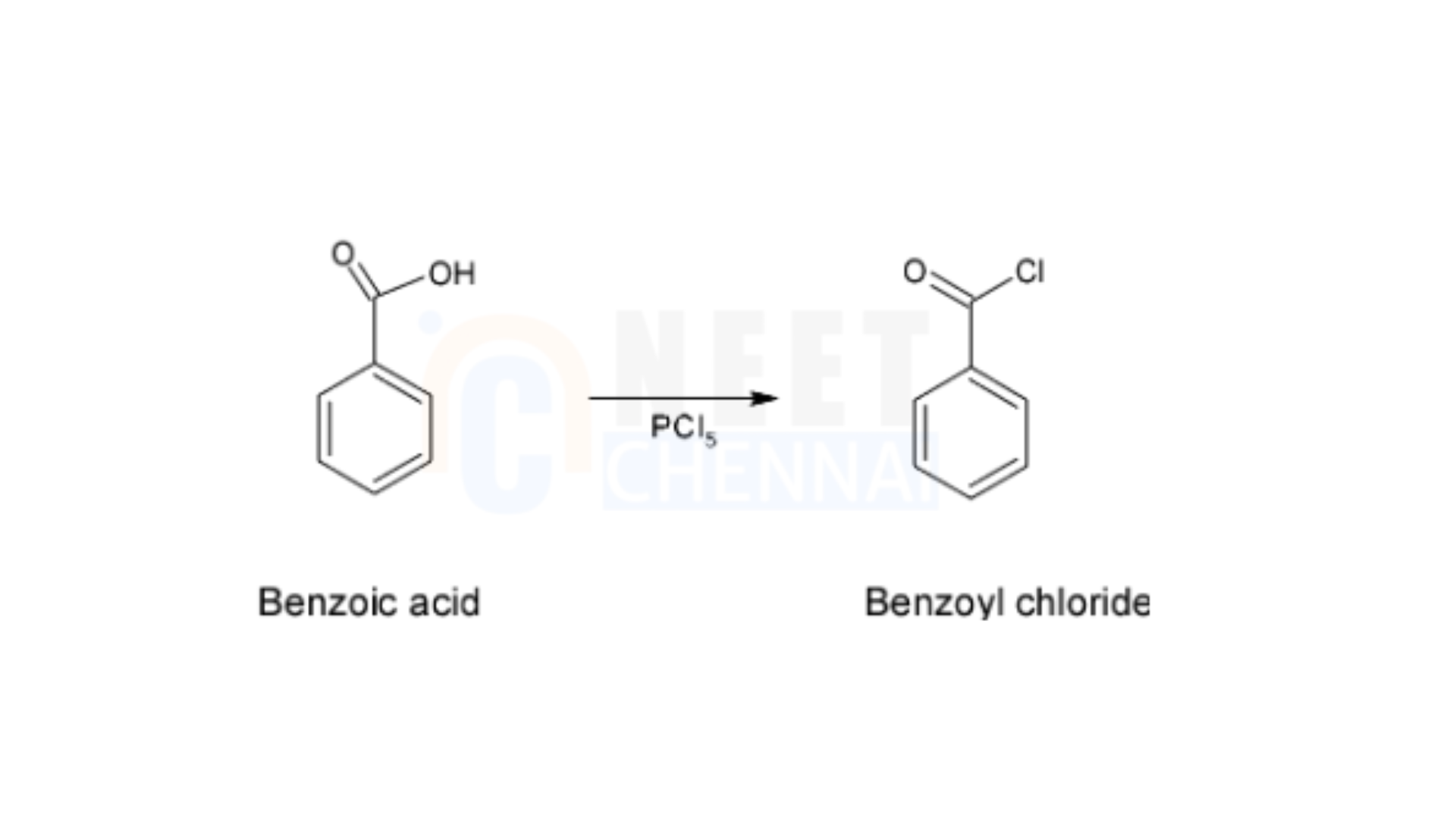
– The reaction starts by converting benzoic acid into benzoyl chloride through treatment with phosphorus pentachloride.
– This reagent changes an acidic group into a chloride group, a process referred to as chlorination. The reaction is illustrated as follows—
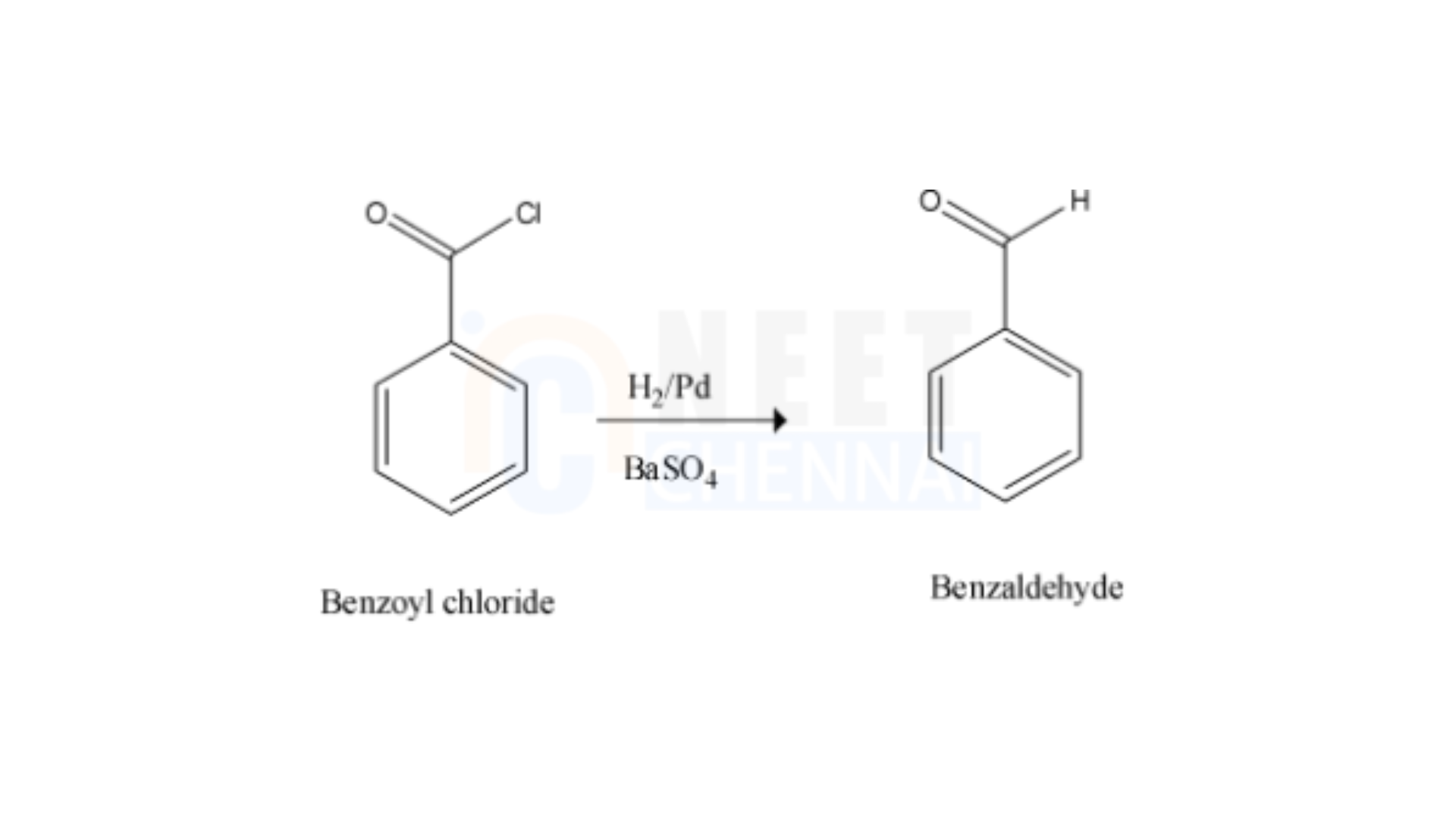
Now, after this conversion, since -Cl group is a better leaving group it easily undergoes a reduction reaction in the presence of Lindlar’s catalyst which has the composition as H2/Pd,BaSO4
As a result, the benzoyl chloride produced undergoes reduction to form the target product when treated with Lindlar’s catalyst. This process is known as Rosenmund’s reduction. The reaction is shown below—
Thus, the whole of the reaction can be given in a single step. This is depicted as given below,
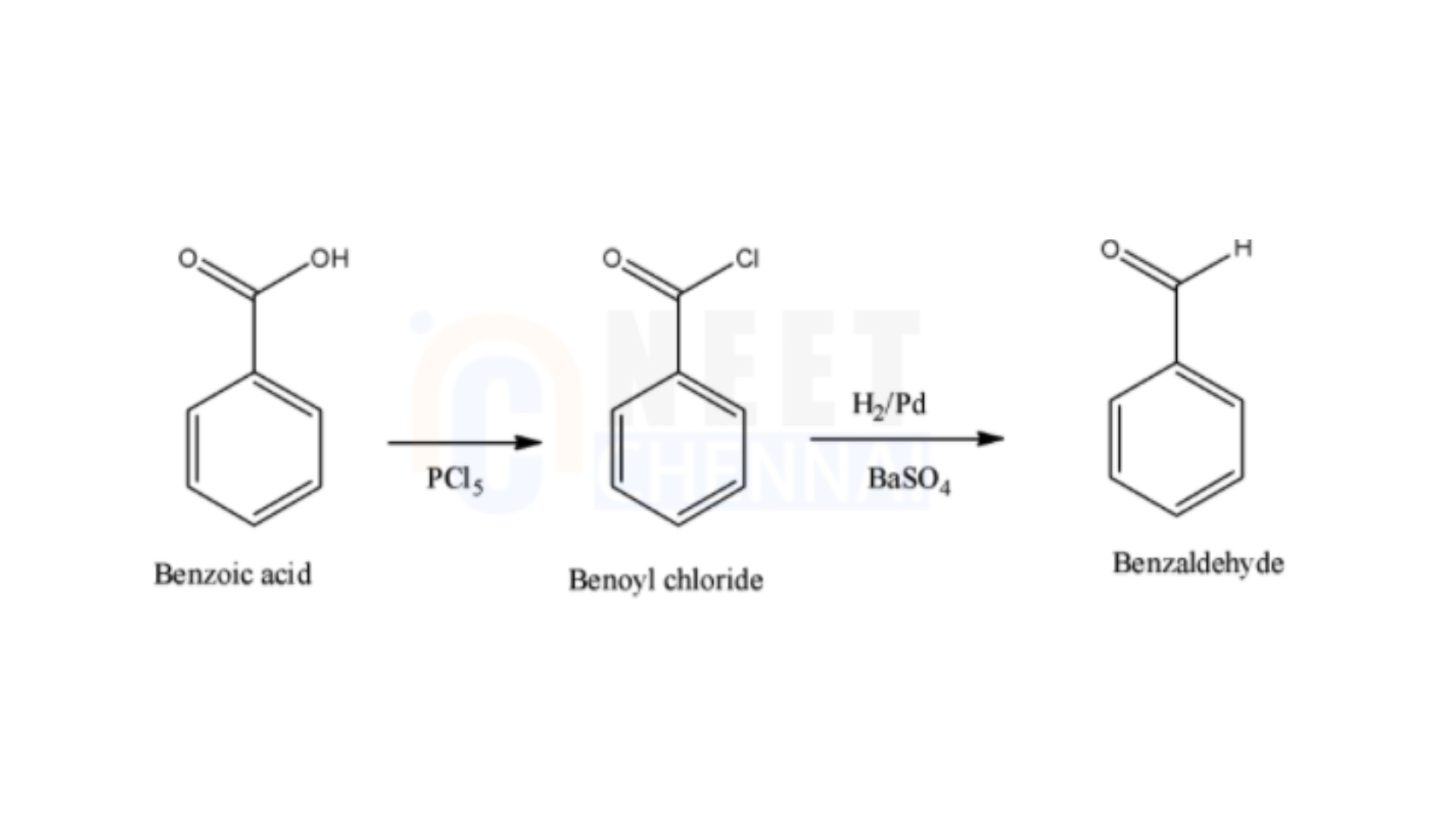 Therefore, the correct answer is benzoic acid is converted to benzaldehyde by chlorination and then followed by the reduction reaction which is a named reduction reaction.
Therefore, the correct answer is benzoic acid is converted to benzaldehyde by chlorination and then followed by the reduction reaction which is a named reduction reaction.
Note: An alternative approach for this reaction involves adjusting the reaction conditions slightly; for example, thionyl chloride can be used in place of phosphorus pentachloride, and Lindlar’s catalyst can be substituted with LiAlH4 followed by PCC or PDC. Don’t get confused if questions like these come up.