a. Give chemical tests to distinguish between:
1. Propanol and propanone
2. Benzaldehyde and acetophenone
B. Arrange the following compounds in order of increasing strength for the given property:
1. Acetaldehyde, acetone, methyl tert butyl ketone (reactivity towards HCN)
2. Benzoic acid, 3,4-dinitrobenzoic acid, 4-methoxybenzoic acid (acid strength)
3. \( \textbf{CH}_3\textbf{CH}_2\textbf{CH(Br)COOH} \), \( \textbf{CH}_3\textbf{CH(Br)CH}_2\textbf{COOH} \), \(
\textbf{CH}_3)_2\textbf{CHCOOH} \) (acid strength)
Hint: Propanol and benzaldehyde contain aldehyde functional groups, while propanone and acetophenone have methyl ketone groups. We need to determine the test used to detect methyl ketones. Electron-donating groups reduce acid strength, whereas electron-withdrawing groups enhance acid strength.
Complete step by step solution:
a. 1. Chemical test to differentiate between propanal and propanone: Propanal contains an aldehyde functional group, while propanone is a methyl ketone. The structures of propanal and propanone are shown as follows:
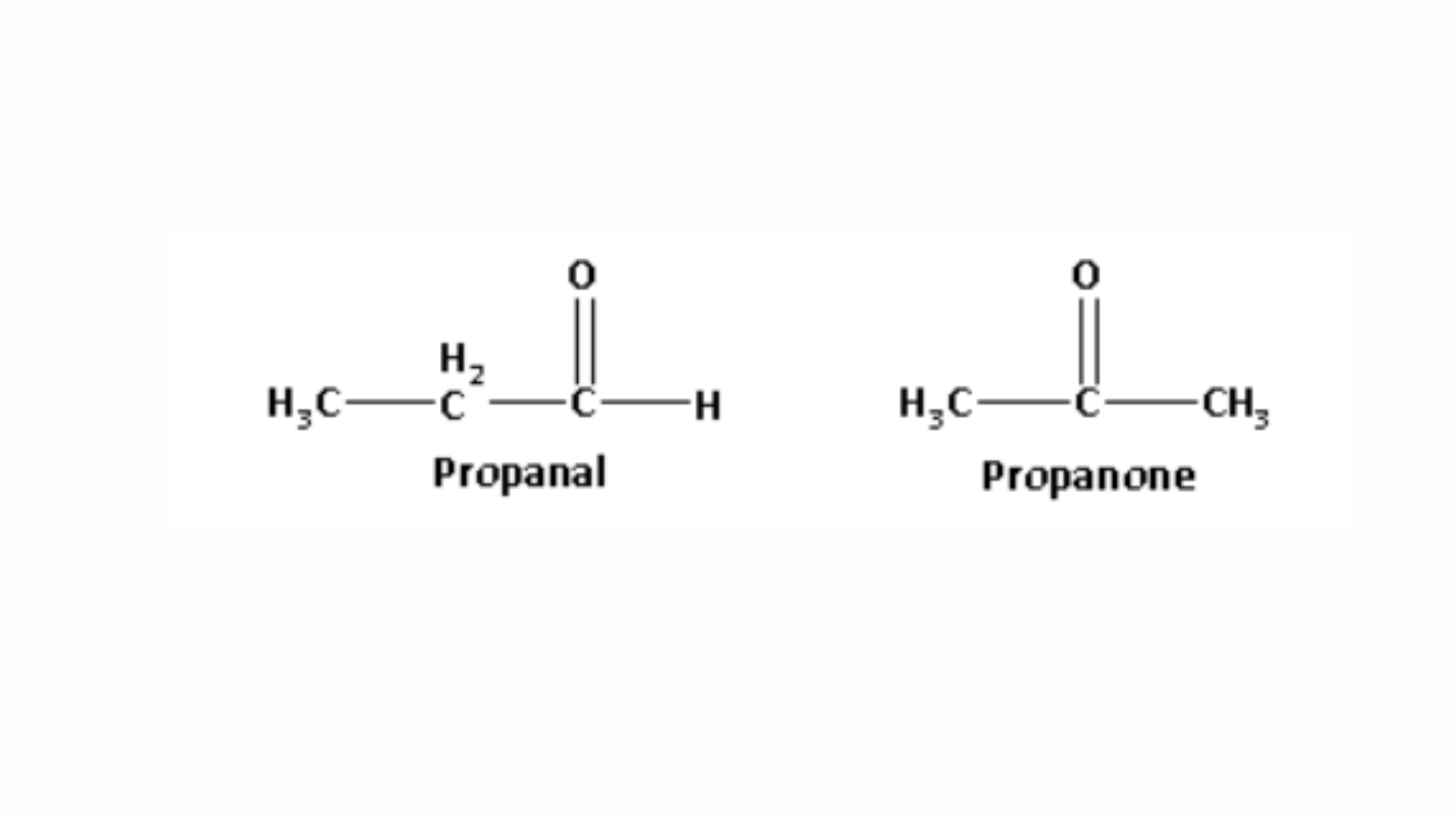
When propanone reacts with sodium hypoiodite, it produces a yellow precipitate of iodoform. The reaction between propanone and sodium hypoiodite is shown as follows:
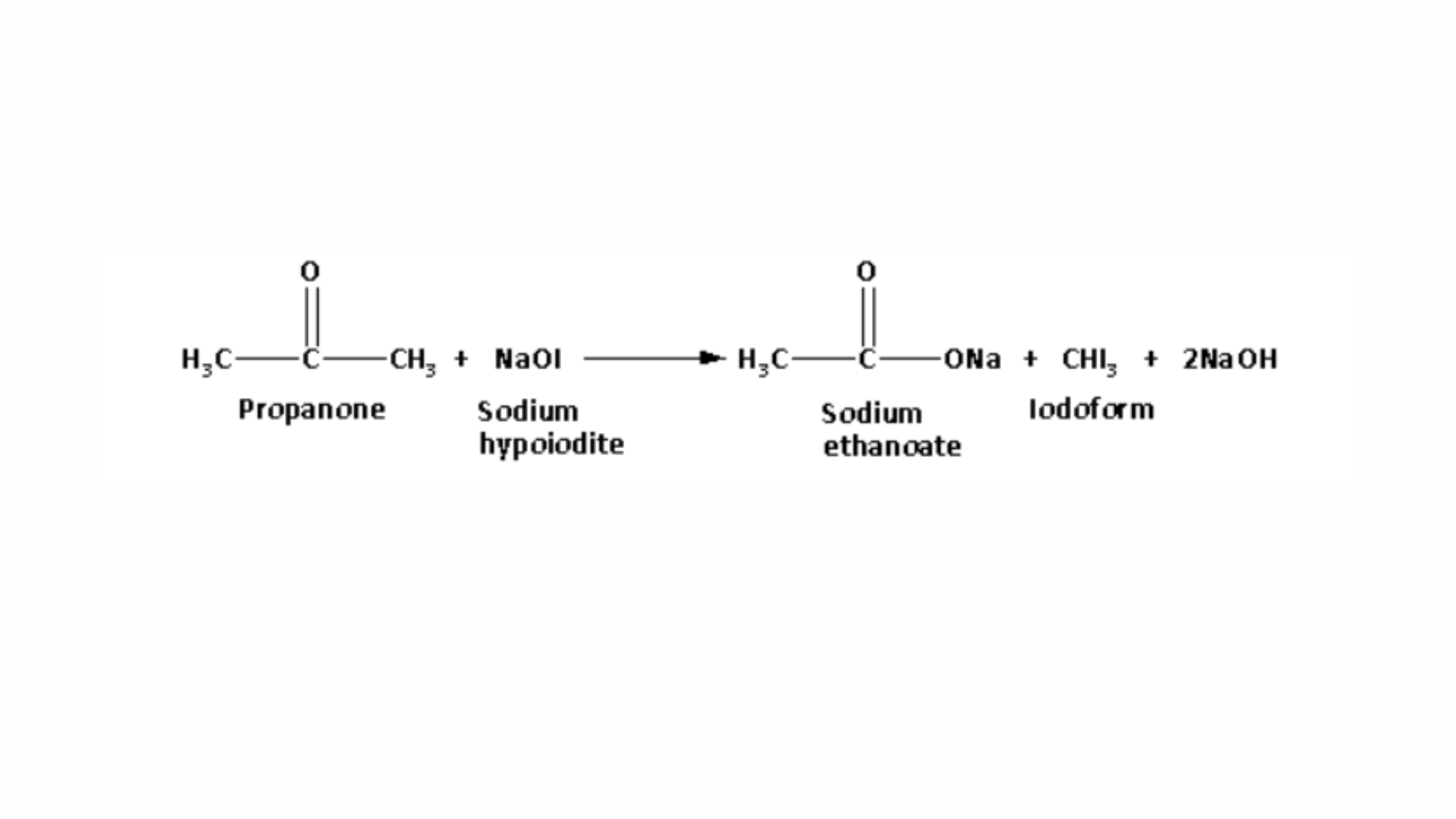
Propanol does not produce a yellow precipitate when reacted with sodium hypoiodite. Therefore, the reaction with sodium hypoiodite serves as the distinguishing test between propanol and propanone, known as the iodoform test. Thus, the iodoform test is the chemical method used to differentiate between propanol and propanone.
2. Chemical test to differentiate between benzaldehyde and acetophenone: Benzaldehyde contains an aldehyde functional group, while acetophenone is a methyl ketone. The molecular structures of benzaldehyde and acetophenone are shown below:
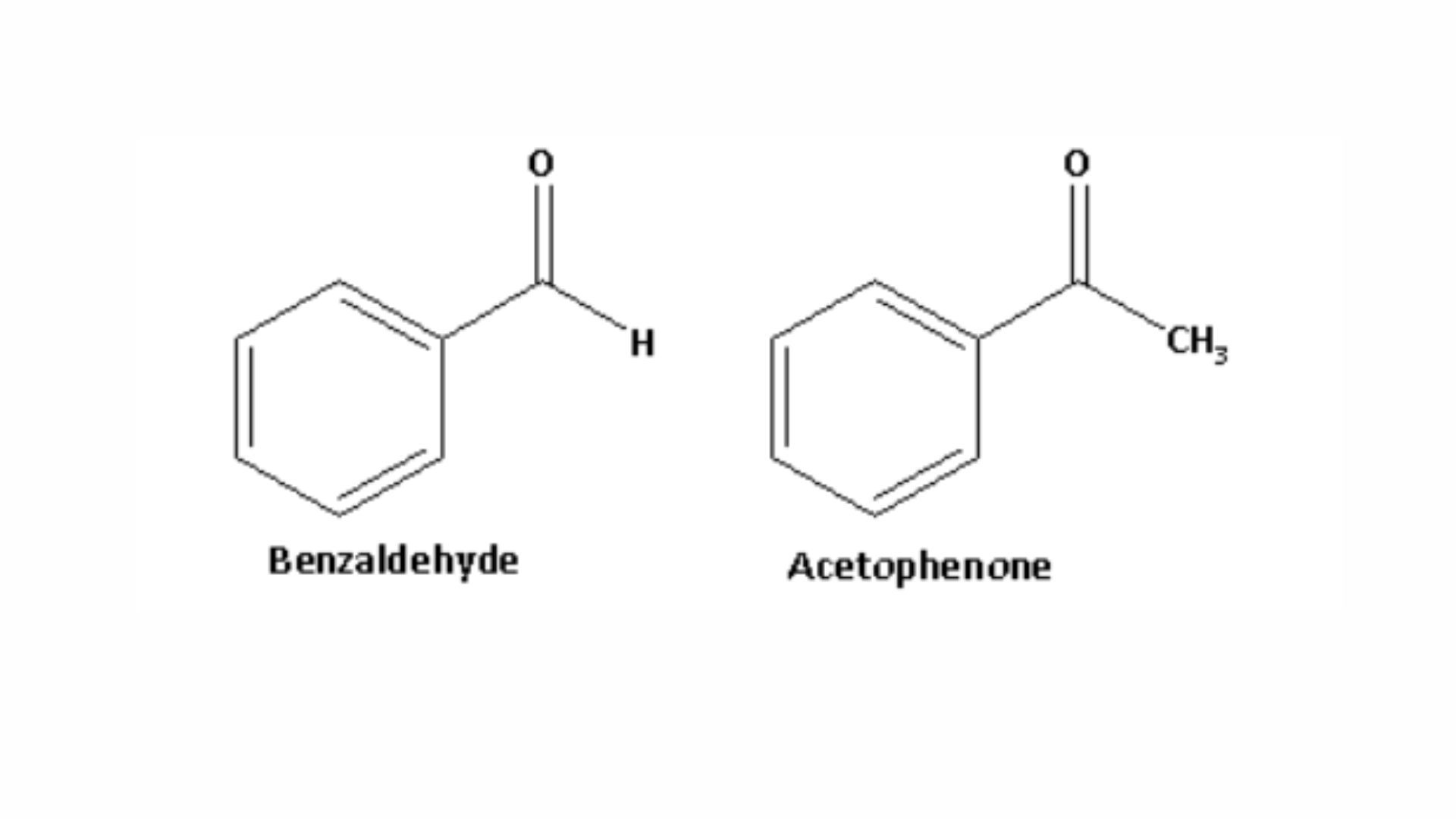
When acetophenone reacts with sodium hypoiodite, it forms a yellow precipitate of iodoform. The reaction between acetophenone and sodium hypoiodite is shown below:
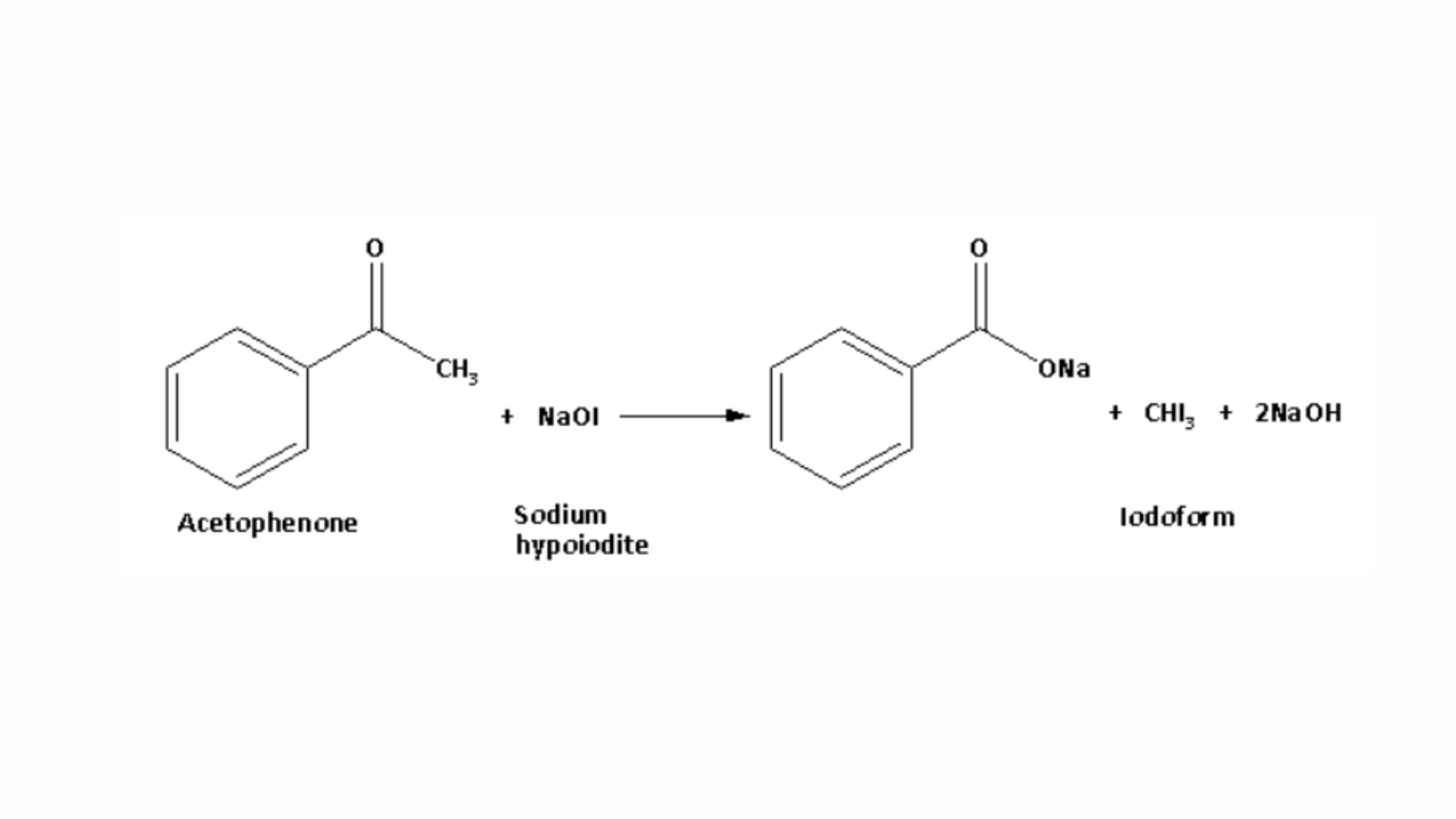
Benzaldehyde does not produce a yellow precipitate when reacted with sodium hypoiodite. Therefore, the reaction with sodium hypoiodite serves as the distinguishing test between benzaldehyde and acetophenone, known as the iodoform test. Hence, the iodoform test is the chemical method used to differentiate between benzaldehyde and acetophenone.
b. 1. Arrange acetaldehyde, acetone, and methyl tert-butyl ketone in order of their reactivity towards HCN, from least to most reactive: The structures of acetaldehyde, acetone, and methyl tert-butyl ketone are as follows:
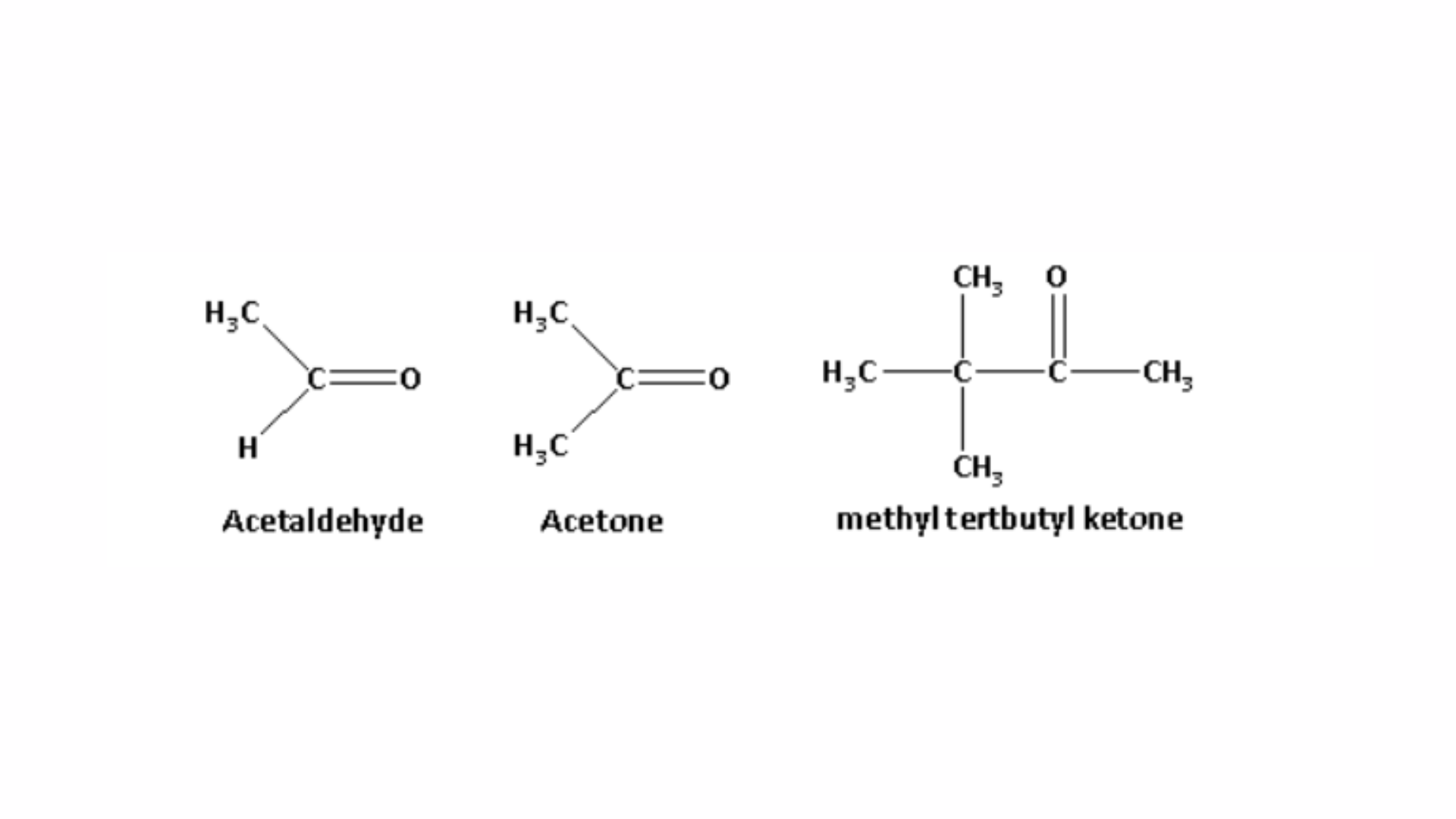
When HCN reacts with a compound, the nucleophile CN− is the species that attacks. The methyl group is an electron-donating group, and as the number of methyl groups increases, the electron density around the carbonyl carbon also increases. The order of electron density is as follows:
Methyl tert-butyl ketone > Acetone > Acetaldehyde.
As the electron density around the carbonyl carbon increases, the compound’s reactivity towards nucleophilic addition decreases. This is because the higher electron density on the carbonyl carbon makes it more negatively charged, which makes it less likely for the negatively charged nucleophile to attack.
Thus, the reactivity of acetaldehyde, acetone, and methyl tert-butyl ketone towards HCN follows this increasing order:
Methyl tert-butyl ketone < Acetone < Acetaldehyde.
2. Arrange benzoic acid, 3,4-dinitrobenzoic acid, and 4-methoxybenzoic acid in order of their acid strength, from weakest to strongest:
The structures of benzoic acid, 3,4-dinitrobenzoic acid, and 4-methoxybenzoic acid are as follows:
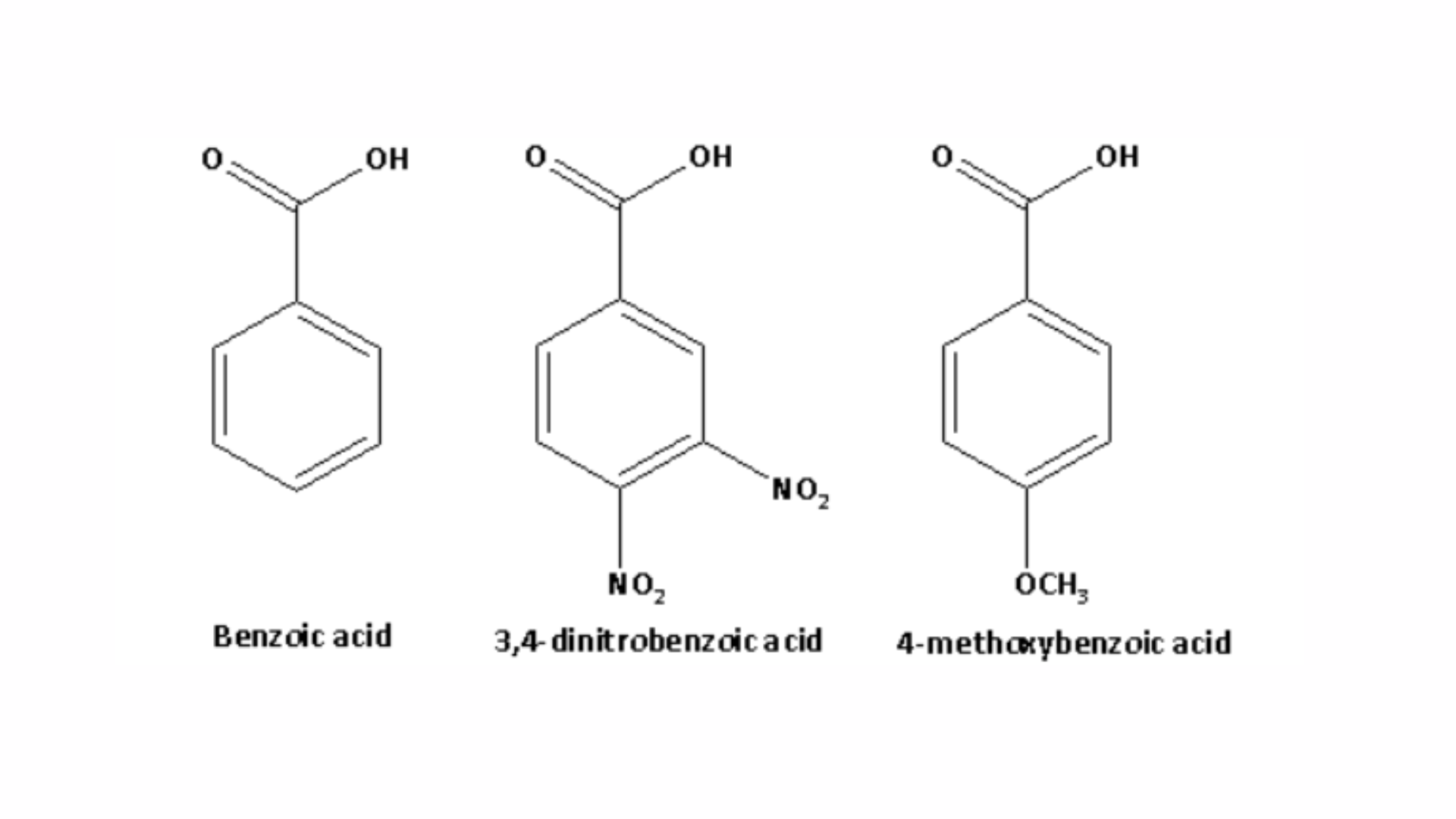
Electron-donating groups reduce acid strength, and the methoxy group is an electron-donating group. On the other hand, electron-withdrawing groups increase acid strength, and the nitro group is an electron-withdrawing group. Therefore, the acid strength of 3,4-dinitrobenzoic acid is greater than that of benzoic acid, while the acid strength of 4-methoxybenzoic acid is weaker than that of benzoic acid.
Thus, the order of acid strength from weakest to strongest for benzoic acid, 3,4-dinitrobenzoic acid, and 4-methoxybenzoic acid is:
4-methoxybenzoic acid < benzoic acid < 3,4-dinitrobenzoic acid.
\( 3. Arrange \text{CH}_3\text{CH}_2\text{CH(Br)COOH}, \text{CH}_3\text{CH(Br)CH}_2\text{COOH}, (\text{CH}_3)_2\text{CHCOOH} \text{ in an increasing order of their acid strengths:}
\)
Electron-donating groups reduce acid strength, and the \( \text{-CH}_3 \) group is an electron-donating group.
Conversely, electron-withdrawing groups enhance acid strength, and the −Br group is an electron-withdrawing group.
\((\text{CH}_3)_2\text{CHCOOH} \) has the lowest acid strength. In \( \text{CH}_3\text{CH}_2\text{CH(Br)COOH} \) , the −Br group is near the acid functional group and in \( \text{CH}_3\text{CH(Br)CH}_2\text{COOH} \), the −Br is away from the acid functional group. As the distance increases, the acid strength decreases.
Thus, \( \text{CH}_3\text{CH}_2\text{CH(Br)COOH} \) is a stronger acid than \( \text{CH}_3\text{CH(Br)CH}_2\text{COOH}. \)
Therefore, the order of acid strength from weakest to strongest is as follows:
\( \text{CH}_3)_2\text{CHCOOH} \) < \( \text{CH}_3\text{CH(Br)CH}_2\text{COOH} \) < \( \text{CH}_3\text{CH}_2\text{CH(Br)COOH} \)
Note:
As the separation between the electron-withdrawing group and the carbonyl carbon increases, the electron-withdrawing effect diminishes, leading to a decrease in acid strength.


