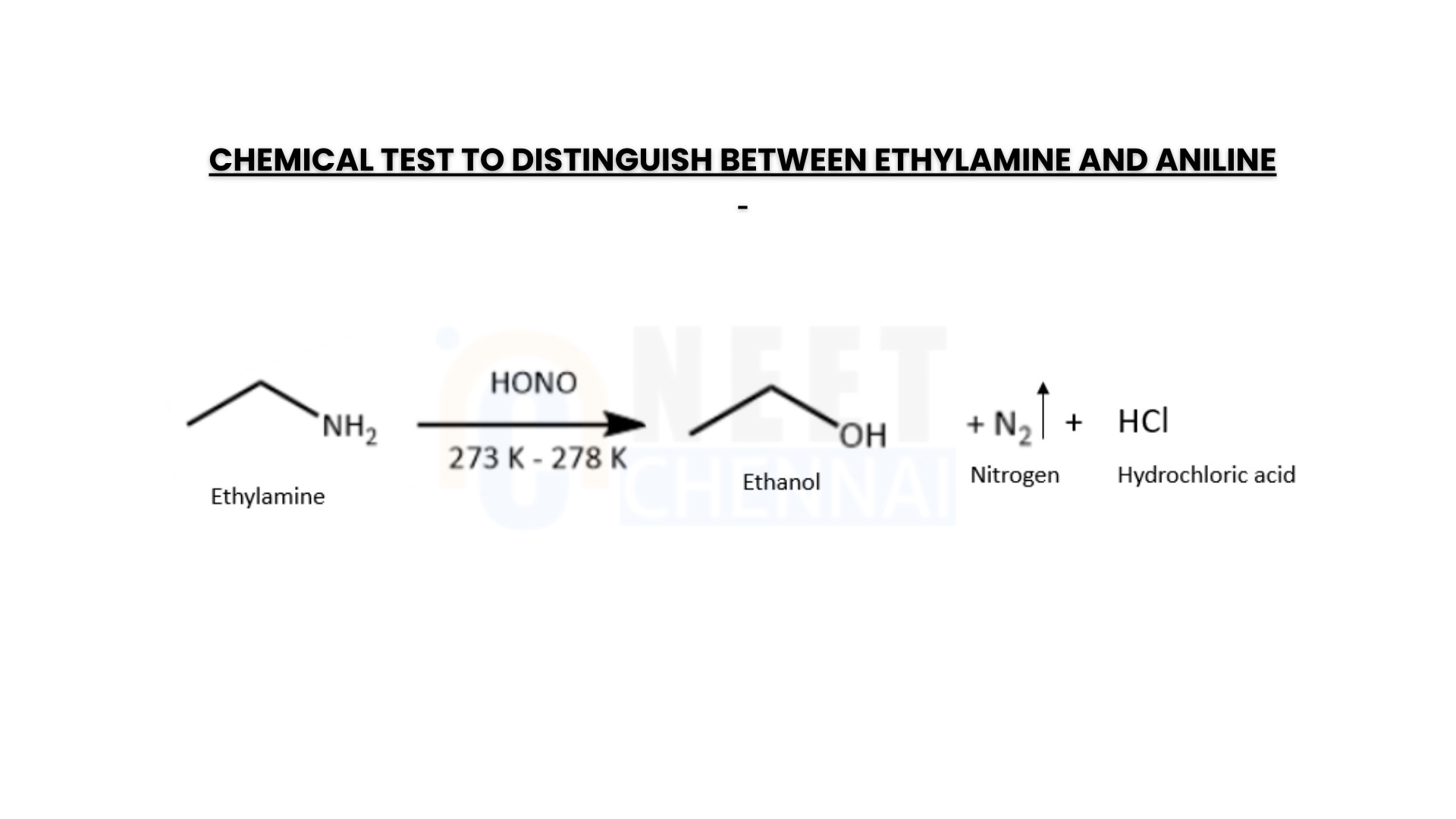
Give one chemical test to distinguish between ethylamine and aniline.
Hint: The reaction products of nitrous acid with aliphatic and aromatic amines exhibit distinct chemical properties. Compounds formed from aliphatic amines release nitrogen gas with bubbling, whereas those from aromatic amines create vivid-colored dyes. Ethylamine is an aliphatic amine, while aniline is an aromatic amine.
Complete answer:
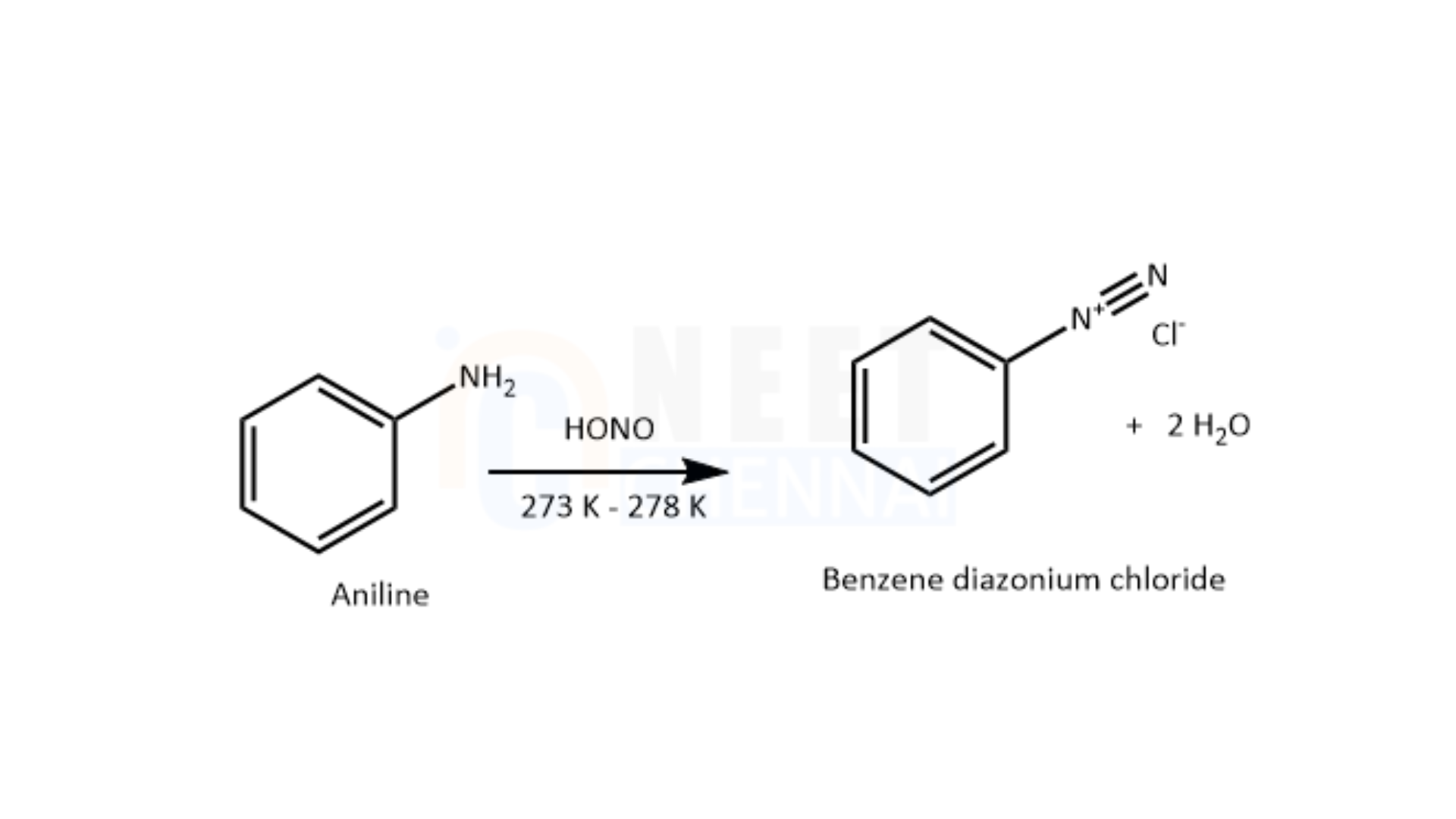
When aromatic amines react with nitrous acid at a low temperature, they produce a diazonium salt. By coupling this diazonium salt with 2-naphthol in the presence of a base, such as an aqueous sodium hydroxide solution, a bright-colored azo dye is formed. However, if nitrous acid is added to aliphatic amines, nitrogen gas is released, causing effervescence. This reaction allows us to differentiate between ethylamine and aniline using the azo dye test. Aniline, being an aromatic amine, reacts with nitrous acid at a low temperature to form benzene diazonium chloride (a type of diazo salt). Nitrous acid is generated in situ by the reaction of sodium nitrite with dilute hydrochloric acid.
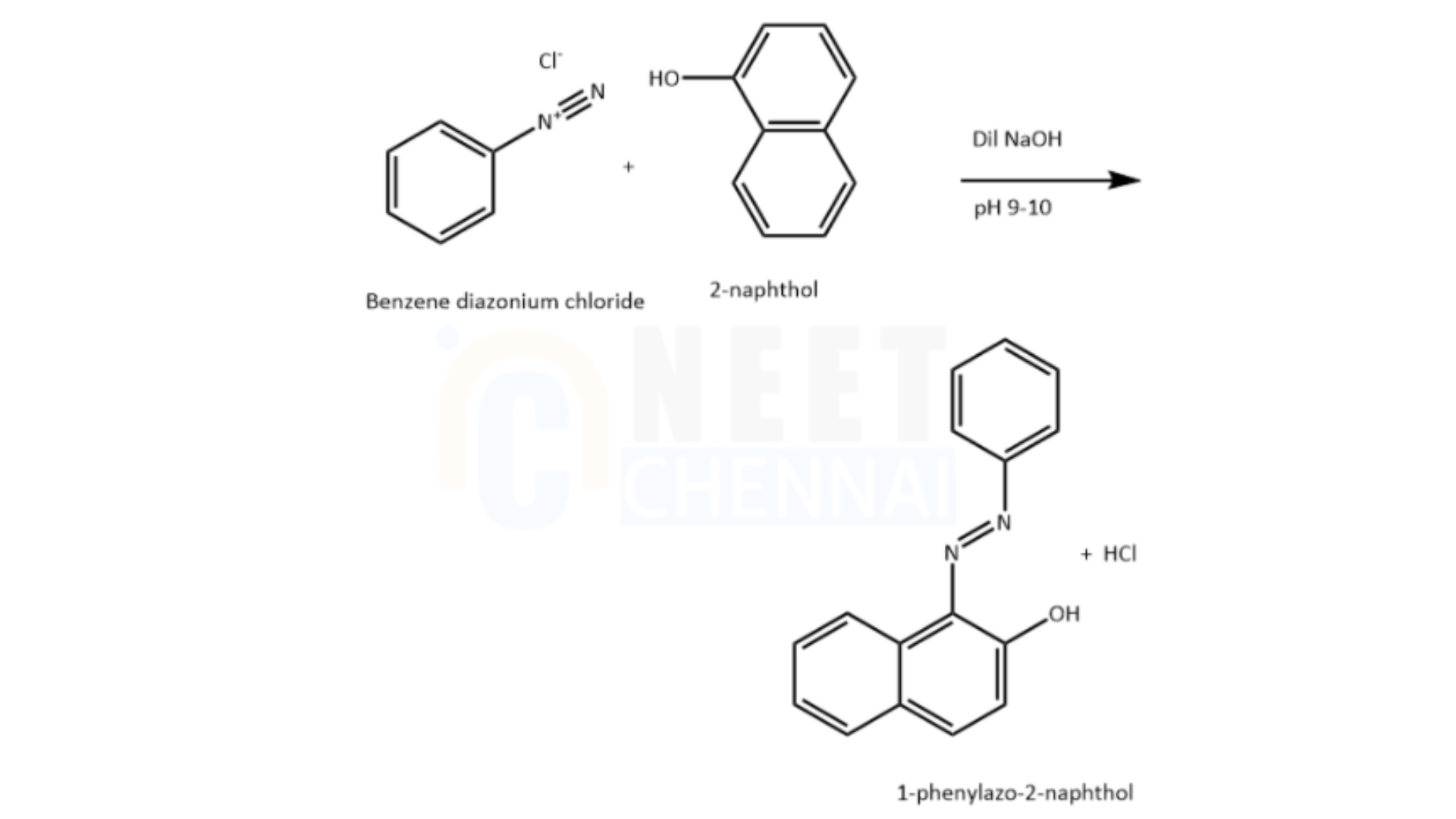
 Benzene diazonium chloride subsequently reacts with an alkaline solution of 2-naphthol, resulting in the formation of an orange-colored dye.
Benzene diazonium chloride subsequently reacts with an alkaline solution of 2-naphthol, resulting in the formation of an orange-colored dye.
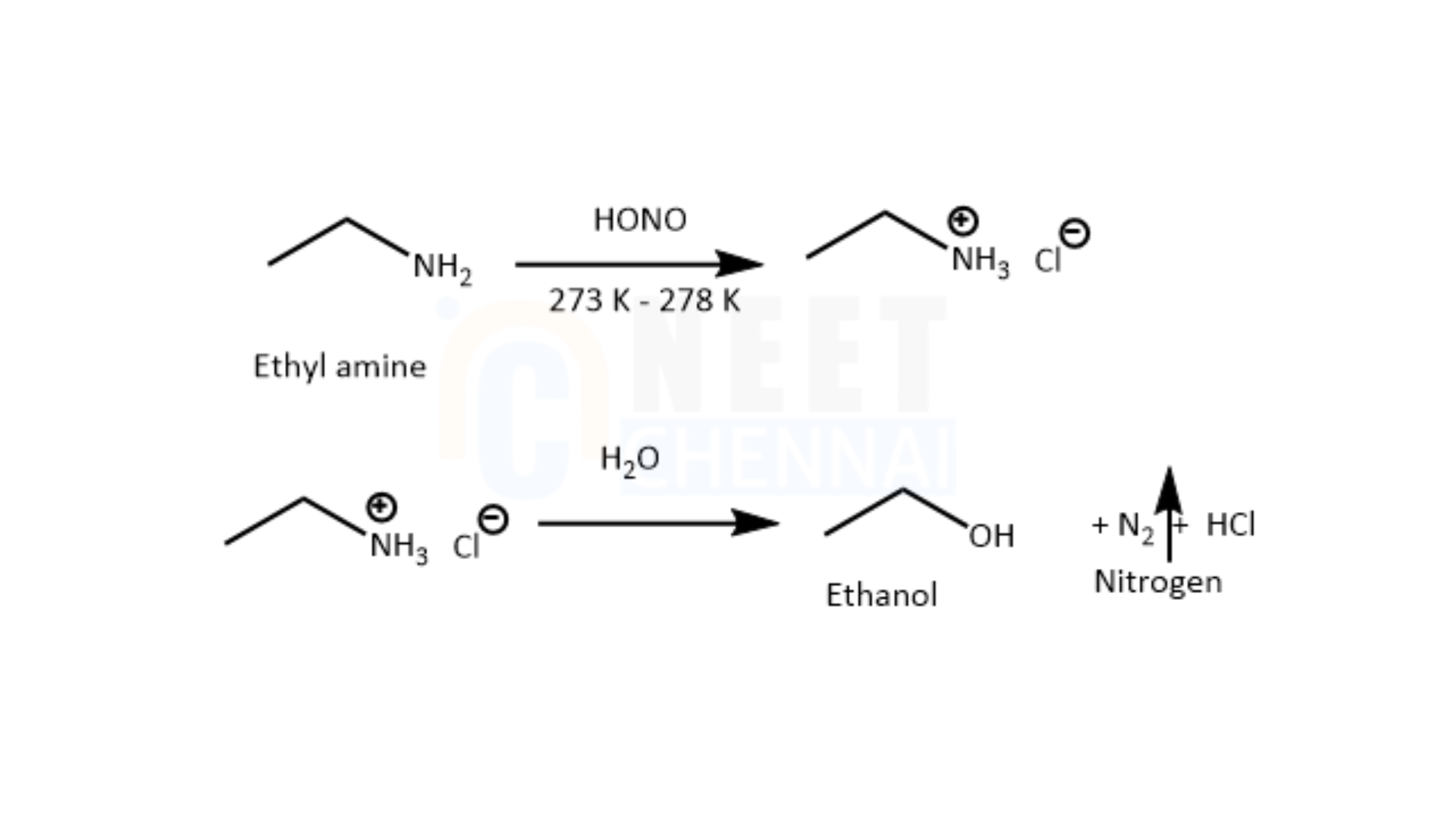
When ethylamine is exposed to similar conditions, rapid bubbling of nitrogen gas is observed.
 The azo dye test is used to distinguish between ethylamine and aniline.
The azo dye test is used to distinguish between ethylamine and aniline.
Note: Azo dyes comprise a large group (approximately 2000 types) of synthetic dyes containing the C – N = N – C bond. These dyes are widely used to add color to leather, textiles, various articles, and some food items. Azo dyes are affordable, simple to use, and offer vibrant, intense colors. Therefore, they are extensively applied in the dyeing industry.