Hint: Ionic crystals are a type of crystal where the ions in the crystal lattice are bound by electrostatic or Coulombic forces of attraction. These crystals exhibit a structured arrangement of ions. They are typically solid and have a high melting temperature.
Complete step by step answer:
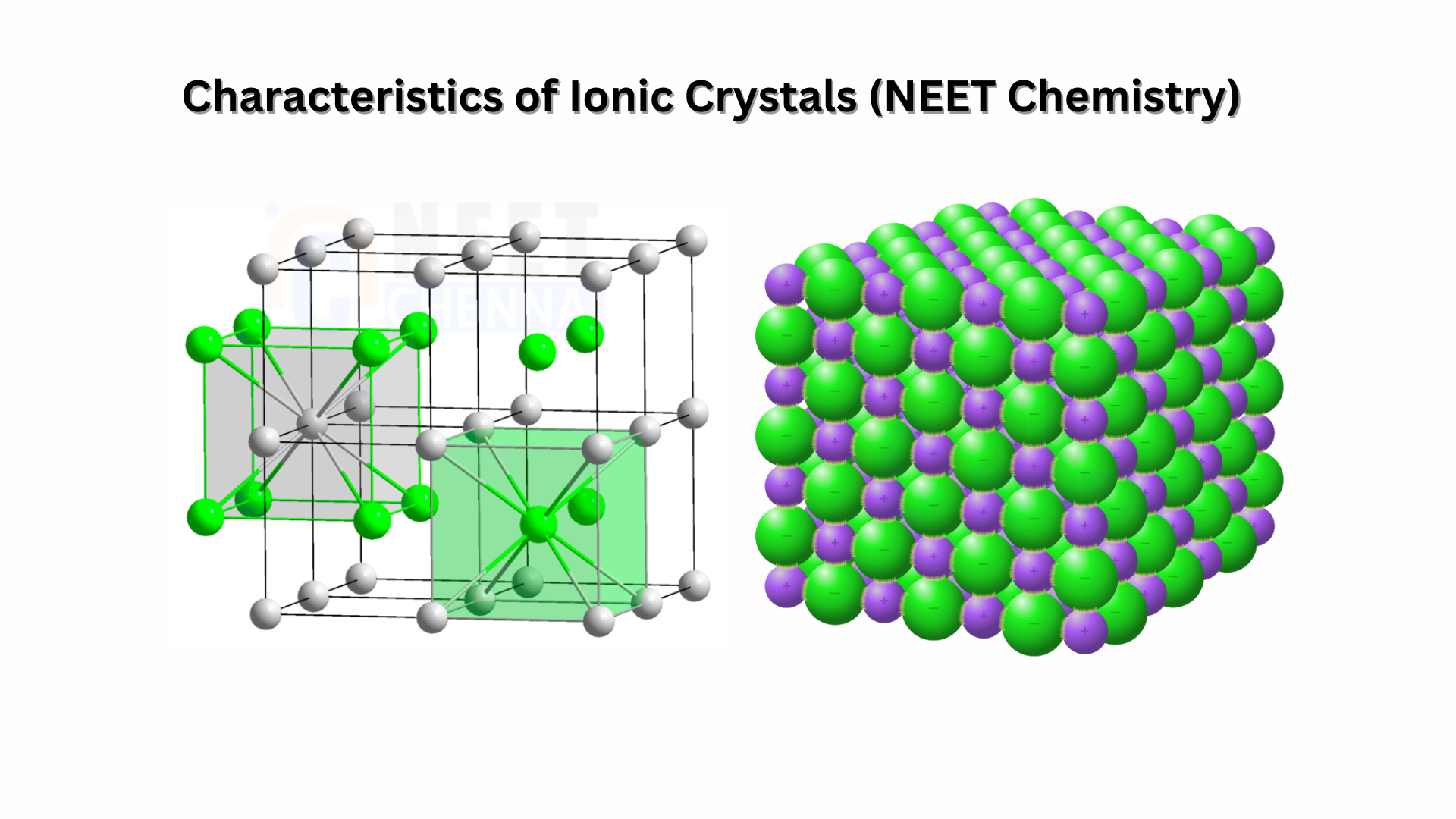
Ionic crystals consist of ions that are bound together by electrostatic forces. Some examples of ionic crystals are alkali halides such as potassium fluoride, potassium chloride, potassium bromide, potassium iodide, and sodium fluoride.
Some key properties of ionic crystals are:
- Ionic crystals consist of charged particles, known as ions. These ions are made up of cations and anions, which are held together by electrostatic forces, also referred to as ionic bonding.
- The arrangement of ions in the crystal lattice is determined by the size or radius ratio between the positive and negative ions.
- The stability of ionic crystals is influenced by lattice energy, which is the amount of energy required to separate the cation and anion in the crystal. For example, when sodium ions and chloride ions combine to form sodium chloride, 787 kJ/mol of energy is released.
The larger lattice energy is due to the stronger Coulombic force of attraction between the charged particles. The energy released during this process helps stabilize the ionic crystal. This is the energy required to break the ionic crystal into its constituent ions.
4. Ionic crystals experience strong interactions between their ions, making them poor conductors of electricity, as they lack free-moving ions.
5. Ionic crystals can absorb infrared radiation, which facilitates the breaking of bonds within the crystal.
5. Ionic crystals are rigid and have a high melting point due to their strong ionic bonds.
Note: It is important to note that ionic crystals have a specific arrangement of ions in their crystal lattice. When in the molten state, ionic crystals can move their ions, making them good conductors of electricity. However, in their solid form, they are poor conductors of electricity.