State the basic postulates of Bohr’s theory of atomic spectra.
Hint: Bohr’s theory focused on the particle-like nature of electrons, proposing that electrons orbit the nucleus in fixed circular paths.
Complete Solution
Bohr integrated ideas from both classical mechanics and early quantum theory to form a model with three main postulates:
Stable Orbits
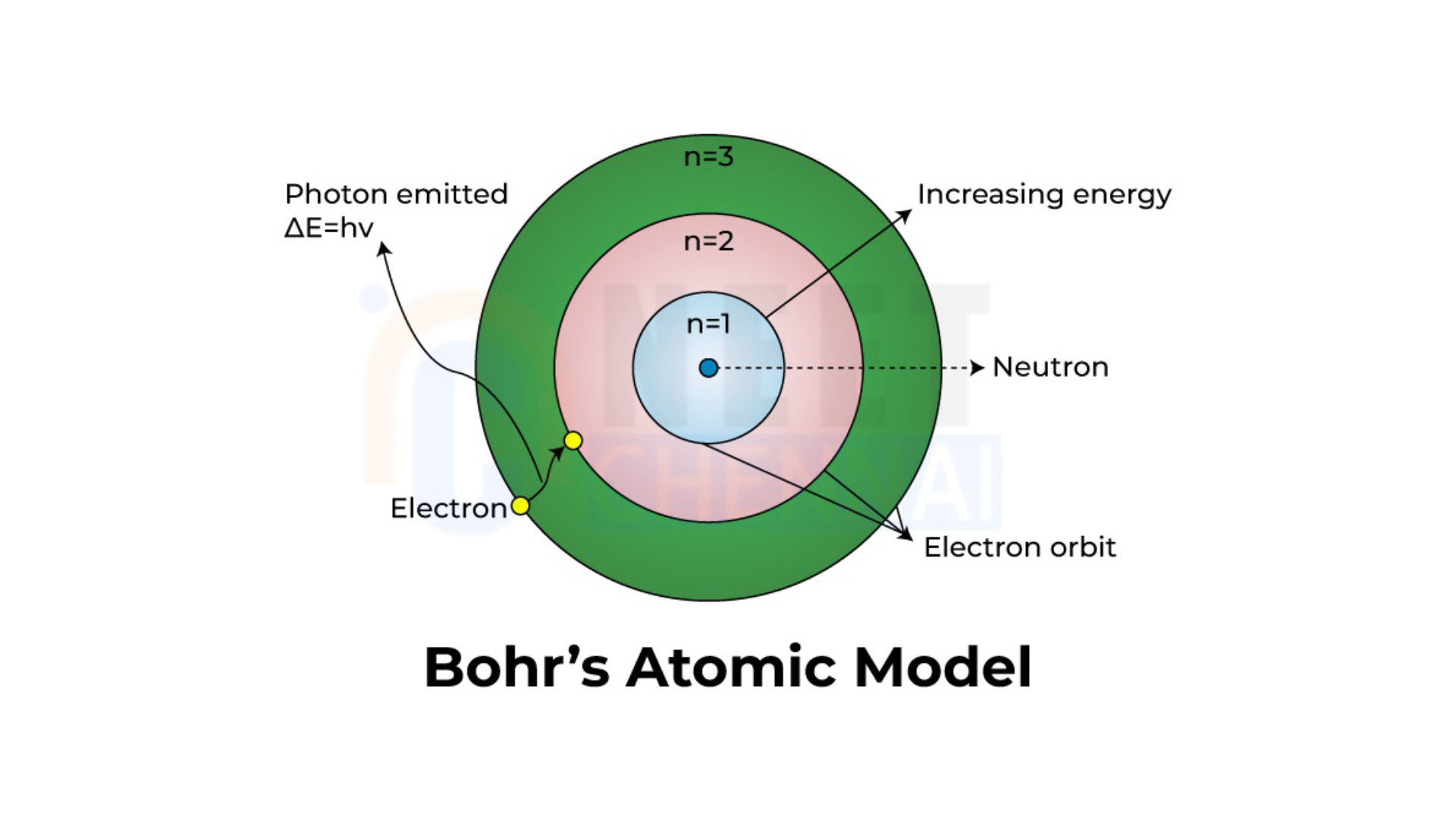
In Bohr’s model, electrons move in specific orbits around the nucleus without emitting energy as they orbit. Each atom has certain allowed orbits, termed “stationary states,” where electrons can exist with fixed energy levels. In these states, electrons do not lose or gain energy.
Quantization of Angular Momentum
Bohr proposed that an electron’s angular momentum in these stable orbits is quantized. This means that it is restricted to certain values and must be a whole-number multiple of \(\frac{h}{2\pi}\), where \(h\) is Planck’s constant. For an electron of mass \(m\) moving with velocity \(v\) in an orbit of radius \(r\), this condition can be expressed as:
\(mvr = n\frac{h}{2\pi}\)
Here, \(n\) is called the “principal quantum number” and can take values like 1, 2, 3, etc. This is known as Bohr’s quantization condition.
Energy Transitions and Photon Emission
According to Bohr, an electron can transition between these orbits. When it moves from a higher-energy orbit to a lower-energy orbit, it emits a photon with energy equal to the difference between the initial and final energy levels. The frequency \(v\) of the emitted photon is given by:
\(v = \frac{E_i – E_f}{h}\)
where \(E_i\) and \(E_f\) are the energies of the initial and final orbits, respectively. This is known as Bohr’s frequency condition.
Note: Bohr’s theory is mainly applicable to hydrogen and hydrogen-like atoms. It does not address the wave-like nature of electrons and does not explain how electrons are distributed across various orbits in multi-electron atoms.