Introduction to Strong and Weak Electrolytes
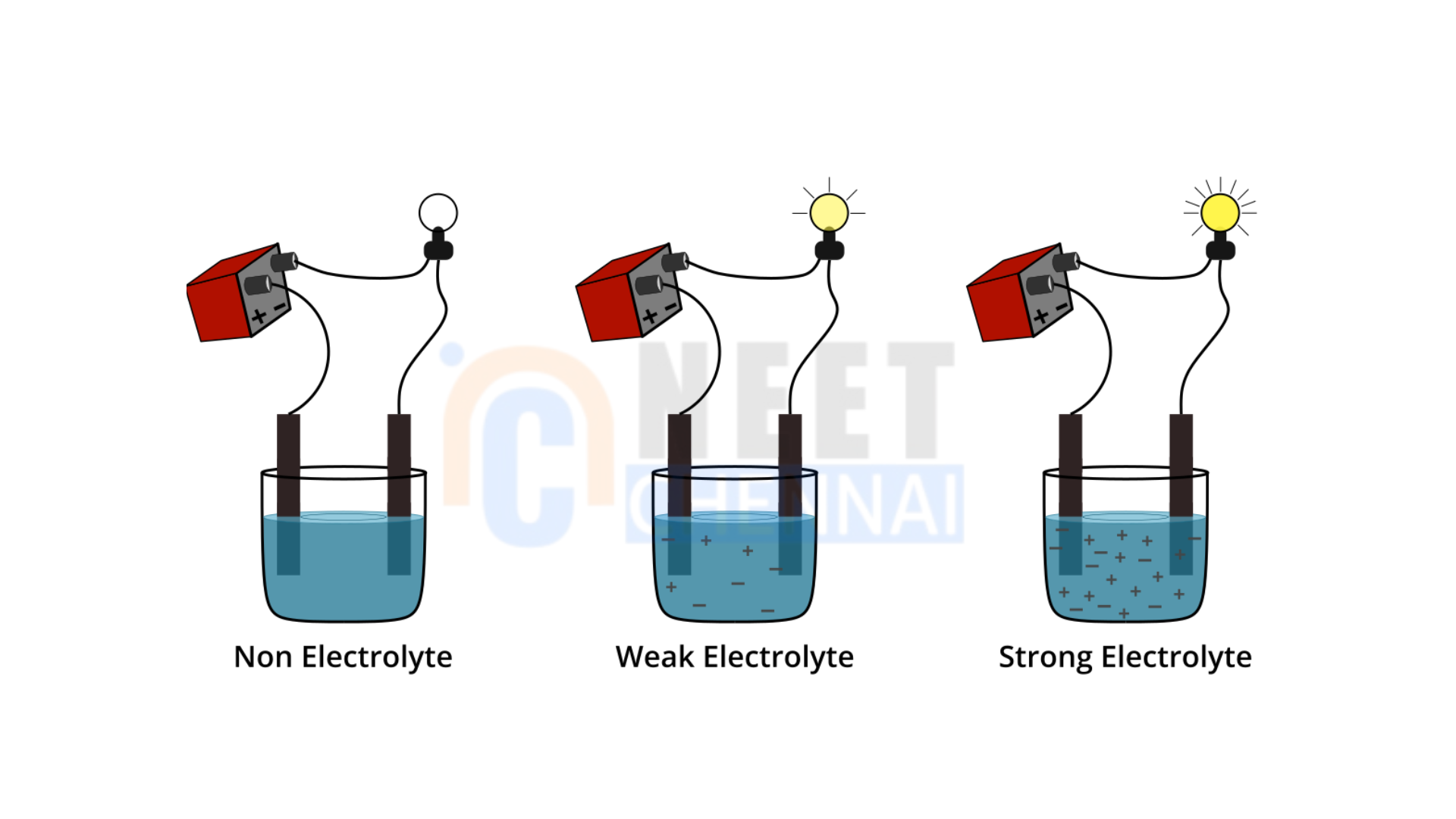
Electrolytes are substances that enable the conduction of electric current when they are dissolved in a solvent, typically water. They are crucial for various biological functions and chemical processes, maintaining the balance of ions necessary for nerve function, muscle contraction, and overall fluid balance. Electrolytes can be categorized into two types: strong electrolytes and weak electrolytes.
Strong Electrolytes are substances that completely ionize in water, breaking down into ions and creating a high concentration of charged particles in the solution. Examples of strong electrolytes include strong acids (like hydrochloric acid and sulfuric acid), strong bases (such as sodium hydroxide and potassium hydroxide), and salts (like sodium chloride and potassium nitrate). These compounds readily release ions, which facilitates efficient electric current conduction.
Weak Electrolytes, in contrast, only partially ionize in solution. This means they dissociate into ions to a lesser degree, resulting in a lower concentration of ions. Common examples include weak acids (like acetic acid) and weak bases (such as ammonia). In solutions of weak electrolytes, a balance exists between their ionized and non-ionized forms.
The Role of Electrolytes in Biological Systems
Electrolytes are vital for maintaining the body’s fluid and ion balance, and they are present in bodily fluids like blood, sweat, and urine. Key electrolytes include sodium (Na+), potassium (K+), calcium (Ca²+), magnesium (Mg²+), chloride (Cl-), bicarbonate (HCO₃-), and phosphate (PO₄³-). These ions are essential for functions such as nerve signalling, muscle contraction, pH balance, and hydration. Imbalances can arise from excessive sweating, vomiting, diarrhoea, or certain health conditions, leading to significant health impacts that may require medical intervention.
Characteristics of Strong Electrolytes
Consider a hot summer day when you might reach for a sports drink to replenish electrolytes. These beverages often contain salts like sodium chloride (NaCl) or potassium chloride (KCl). When dissolved in water, these salts completely dissociate into sodium (Na+) and chloride (Cl-) ions, resulting in a solution with a high ion concentration that efficiently conducts electricity.
Characteristics of Weak Electrolytes
Imagine adding a few drops of acetic acid (CH₃COOH) to water while preparing a salad dressing. Acetic acid is a weak electrolyte, which only partially ionizes when dissolved. In this case, it produces hydrogen ions (H+) and acetate ions (CH₃COO-). Unlike strong electrolytes, weak electrolytes only produce a limited number of ions, leading to a lower overall concentration in solution.
Applications of Strong and Weak Electrolytes
Strong Electrolytes:
- Electroplating: Used to deposit metal coatings onto surfaces.
- Battery Electrolytes: Facilitate ion movement in batteries, allowing for electric current flow.
- Conductivity Testing: Employed in conductivity meters for accurate measurements.
- Chemical Synthesis: Reactivity in chemical reactions is enhanced by strong electrolytes.
- Industrial Processes: Utilized in processes like metal extraction and wastewater treatment.
Weak Electrolytes:
- pH Regulation:Help maintain stable pH levels in various systems.
- Buffer Solutions:Essential for stabilizing pH in biological and biochemical applications.
- Pharmaceuticals: Improve solubility and bioavailability in drug formulations.
- Acid-Base Reactions: Facilitate controlled and reversible chemical reactions.
- Biological Functions: Play critical roles in osmotic balance, pH regulation, and enzymatic reactions.
Comparison of Strong and Weak Electrolytes
| Sr. No. | Category | Strong Electrolyte | Weak Electrolyte |
|
1 |
Definition | Almost completely dissociates in solution | Partially dissociates in solution |
|
2 |
Ionization | Nearly complete ionization | Partial ionization |
|
3 |
Conductivity | High conductivity | Lower conductivity |
|
4 |
Dissociation | Substantial dissociation into ions | Limited dissociation into ions |
|
5 |
Examples | Strong acids (HCl, H₂SO₄), strong bases (NaOH, KOH), salts (NaCl, KNO₃) | Weak acids include substances like acetic acid and carbonic acid, while weak bases comprise compounds such as ammonia. Additionally, certain salts, like ammonium acetate, also fall into this category. |
|
6 |
Equilibrium | No equilibrium; complete ionization | Exists in equilibrium between ions and uncharged molecules
|
|
7 |
Ions in Solution | High concentration of ions | Lower concentration of ions |
|
8 |
Electrical Conductivity | Greater ability to conduct electricity | Lower ability to conduct electricity |
Summary
Electrolytes are key players in conducting electricity in solutions, crucial for numerous physiological processes. They are categorized as strong or weak based on their ionization levels. Strong electrolytes fully dissociate into ions, resulting in high electrical conductivity, while weak electrolytes only partially dissociate, leading to lower conductivity. Strong electrolytes are widely used in applications like electrolysis and battery systems, while weak electrolytes are important in pH regulation, buffering, and biological systems.